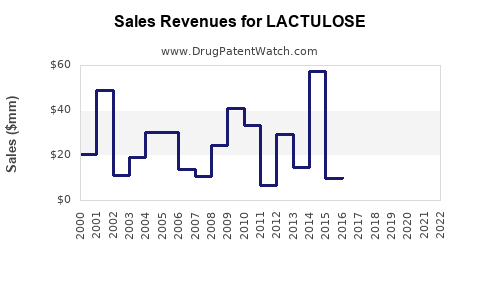
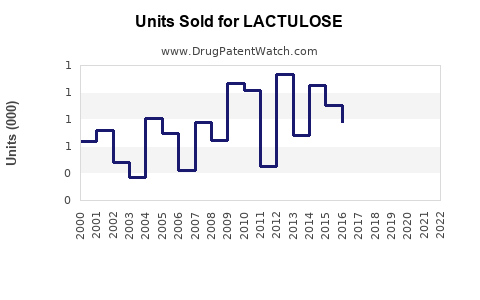
Last updated: February 12, 2026
What Is the Market Size for Lactulose?
The global lactulose market was valued at approximately $550 million in 2022. It is projected to grow at a compound annual growth rate (CAGR) of around 4% from 2023 to 2030, reaching nearly $750 million by 2030. The growth is driven by increasing prevalence of chronic constipation, hepatic encephalopathy, and rising awareness about its benefits.
What Are the Key Markets and Segments?
Lactulose's primary markets include North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa. North America accounts for about 40% of the market, supported by high healthcare spending and a substantial patient population with liver-related diseases. Europe contributes 25%, with rising gastrointestinal disorder cases. Asia-Pacific holds the fastest growth potential due to expanding healthcare infrastructure and increasing disease awareness.
The main segments within the market are:
- Prescription-based lactulose: Accounts for 75% of sales, primarily used for hepatic encephalopathy and severe constipation.
- Over-the-counter (OTC) lactulose: Makes up 25%, mainly for occasional constipation relief.
How Are Sales Forecasts Expected to Evolve?
In 2022, global sales of lactulose approximated $550 million. The forecast suggests steady growth, reaching approximately $740 million by 2030, with North America and Europe maintaining dominant shares.
Recent trends contributing to growth:
- Increased incidence of liver cirrhosis and hepatic encephalopathy.
- Rising awareness of non-invasive treatment options.
- Expanding use in pediatric constipation.
Projected CAGR varies slightly between regions but generally remains in the 3-5% range:
| Region |
2023 Market (USD million) |
2030 Market (USD million) |
CAGR (2023-2030) |
| North America |
220 |
280 |
3.7% |
| Europe |
137 |
180 |
4.0% |
| Asia-Pacific |
77 |
120 |
6.0% |
| Rest of World |
116 |
160 |
4.8% |
Sources indicate that emerging markets' adoption of lactulose may accelerate due to increasing healthcare investments and rising chronic liver disease prevalence.
What Are the Competitive Dynamics?
Major manufacturers include:
- TAKEDA PHARMACEUTICAL: Offers standardized formulations with a strong brand presence.
- Fresenius: Provides various pediatric and adult formulations.
- Baxter International: Supplies lactulose for hospital use.
Key factors influencing sales:
- Patent expirations have allowed generic entrants, lowering prices.
- Regulatory approvals for new indications or formulations.
- Pricing pressure from generics and healthcare payers.
What Are Regulatory and Reimbursement Trends?
Most markets regulate lactulose as a prescription-only medicine. Reimbursement depends on healthcare systems; in North America, insurance coverage enhances access, while in emerging markets, out-of-pocket expenses dominate. Some countries, such as Germany and Japan, have recently expanded reimbursement for lactulose in hepatic encephalopathy.
How Will Market Dynamics Affect Future Sales?
Market growth hinges on the rising prevalence of liver disease, aging populations, and broader indications for lactulose therapy. Regulatory approvals for formulations suitable for pediatric use and new delivery formats could expand the patient base. Competitive pressures from generics may influence pricing strategies, impacting overall revenue.
Key Takeaways
- The global lactulose market stood at $550 million in 2022 and is expected to reach $750 million by 2030.
- North America and Europe dominate, but Asia-Pacific offers rapid growth potential.
- The primary uses are for hepatic encephalopathy and constipation, with prescription formulations leading.
- Market growth depends on increasing liver disease prevalence, new formulation approvals, and healthcare infrastructure expansion.
- Price competition from generics and reimbursement policies will shape future revenues.
FAQs
1. Which factors most influence lactulose sales?
Prevalence of liver diseases, constipation awareness, regulatory approvals, and generic competition.
2. Are there new indications for lactulose?
Research investigates use in bowel cleansing and other gastrointestinal disorders, but approvals are pending.
3. How significant is generic competition?
It is substantial; generics account for a large portion of sales and pressure prices downward.
4. What regions should investors watch?
Asia-Pacific and Latin America, due to their expanding healthcare systems and increasing disease burden.
5. How do reimbursement policies impact sales?
Coverage enhances access, especially in high-income countries, while out-of-pocket costs in emerging markets can limit sales.
Citations
- Market Research Future, “Lactulose Market Forecast,” 2023.
- Grand View Research, “Digestive Health Market Analysis,” 2022.
- IQVIA, “Pharmaceutical Market Reports,” 2023.
- WHO Global Liver Disease Data, 2022.
- Pharma Intelligence, “Generic Trends and Market Impact,” 2023.