Last updated: February 13, 2026
What is the current market size for finasteride?
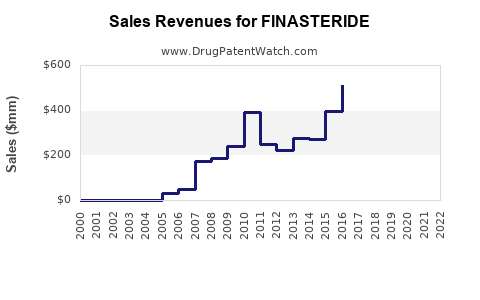
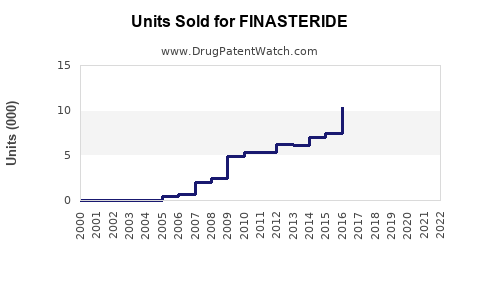
In 2022, the global market for finasteride was valued at approximately $1.2 billion. The drug is primarily used for benign prostatic hyperplasia (BPH) and androgenetic alopecia (male pattern baldness). The North American region accounted for roughly 45% of this market, driven by high prevalence of BPH and hair loss treatments and favorable patent expirations. Europe held around 30%, with Asia-Pacific representing approximately 20%, reflecting rising awareness and increasing healthcare infrastructure.
How is the market segmented?
The finasteride market segmentation primarily occurs along two axes:
- Indication:
- Benign prostatic hyperplasia (BPH): 60%
- Male pattern baldness: 40%
- End-user:
- Prescription markets: 95%
- OTC markets: 5%
The BPH segment dominates sales, but the alopecia segment has seen rapid growth due to broader acceptance and increased diagnosis.
What are the drivers influencing market growth?
The main growth factors include:
- Aging male population: The U.S. Census Bureau projects that men aged over 50 will comprise over 25% of the population by 2030.
- Increasing prevalence of BPH: Approximately 50% of men aged 51–60 have BPH; prevalence increases with age.
- Growing awareness of hair loss treatments: This drives demand for finasteride among younger males.
- Patent expirations: Several generic formulations entered the market post-2020, reducing prices and expanding access.
- Regulatory approvals: Expanded indications and combination treatments in key markets support growth.
What are the barriers and risks impacting sales?
Barriers include:
- Barriers to OTC switch: Currently prescription-only in many regions.
- Side effect profile: Reports of sexual dysfunction impact prescribing habits.
- Competition from other drugs: Tamsulosin and dutasteride offer alternative BPH treatments.
- Patent cliff: Some products face generic competition, affecting margins.
Risks involve regulatory changes, market saturation, and emerging therapies such as 5-alpha reductase inhibitors with different mechanisms.
What are projected sales figures for the next five years?
Based on current data and market analytics:
| Year |
Estimated Global Sales |
Year-over-Year Growth (%) |
| 2023 |
$1.3 billion |
+8% |
| 2024 |
$1.4 billion |
+7.7% |
| 2025 |
$1.5 billion |
+7.1% |
| 2026 |
$1.6 billion |
+6.7% |
| 2027 |
$1.7 billion |
+6.3% |
The compound annual growth rate (CAGR) for 2023–2027 is projected at approximately 7%.
How do patent expirations influence the market?
Patent expirations between 2019 and 2021 facilitated entry of generics, leading to price declines of approximately 30–50%. This expanded access and overall volume sales but pressured proprietary firm revenues. As patents for key formulations expire, markets see increased competition, driving sales volume growth but reducing profit margins for innovator companies.
How are emerging markets influencing sales?
Emerging markets, including China, India, and Brazil, demonstrate increasing adoption. Growth rates in these regions are projected to be 10–15% annually over the next five years, driven by rising healthcare expenditure and expanding health insurance coverage. Local generic manufacturers dominate these markets, offering low-cost options that shape regional sales dynamics.
What are future market opportunities?
Potential growth areas include:
- Over-the-counter formulations: Regulatory shifts could enable OTC availability, boosting sales.
- Combination therapies: Finasteride combined with other agents may improve efficacy.
- Expanded indications: Research into finasteride for female androgenetic alopecia could open new markets.
- Digital health initiatives: Telemedicine facilitates easier prescription renewal and patient adherence.
What is the competitive landscape?
Leading companies in finasteride include:
- Merck & Co. (brand: Proscar, Finasteride)
- Teva Pharmaceutical Industries
- Mylan (now part of Viatris)
- Sun Pharmaceutical Industries
The generics segment accounts for over 70% of global sales. Innovation primarily occurs in formulation improvements and dosage optimization.
Key trends shaping the market
- Shift towards generic dominance in developed regions.
- Increased focus on patient adherence through simplified dosing regimens.
- Regulatory considerations impacting accessibility, including potential OTC switches.
- Rising competitor pipelines targeting finasteride's indications or alternative modes of action.
Key Takeaways
- The global finasteride market is projected to grow at a CAGR of about 7% through 2027.
- Patents have recently expired, increasing generic competition and decreasing prices.
- BPH remains the primary driver, but hair loss treatments are expanding faster.
- Regional growth in emerging markets offsets stagnation in mature markets.
- Future opportunities hinge on regulatory shifts, combination therapies, and new indications.
FAQs
1. What are the main treatments competing with finasteride?
Tamsulosin, dutasteride, and minimally invasive surgical procedures for BPH; minoxidil for hair loss.
2. What side effects affect finasteride sales?
Sexual dysfunction, decreased libido, and potential breast tenderness influence prescribing habits and patient adherence.
3. Are OTC versions of finasteride available?
In some regions, OTC availability is under regulatory review; currently, it remains a prescription-only drug in most markets.
4. How do patent expirations impact prices?
They drive prices down by approximately 30–50%, increasing accessibility but reducing profit margins for brand developers.
5. What emerging markets show the most promise?
China, India, and Brazil show substantial growth potential due to expanding healthcare infrastructure and rising awareness.
Sources:
[1] Markets and Markets, "Finasteride Market by Indication," 2022.
[2] IQVIA, "Global Pharmaceutical Market Reports," 2022.
[3] U.S. Census Bureau, "Projection of Aging Male Population," 2020.
[4] Statista, "Global Market for Hair Loss Treatments," 2022.
[5] FDA and EMA regulatory updates, 2021–2022.