Share This Page
Drug Sales Trends for EXALGO
✉ Email this page to a colleague
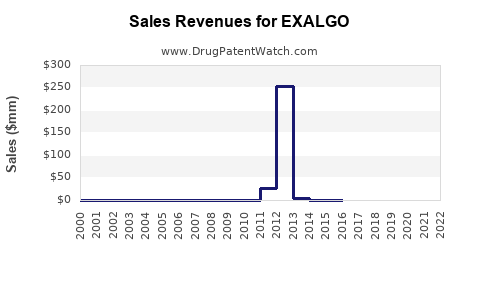
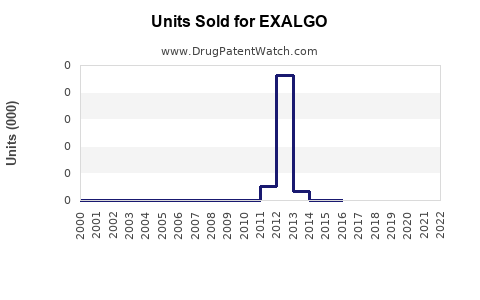
Annual Sales Revenues and Units Sold for EXALGO
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| EXALGO | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| EXALGO | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| EXALGO | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| EXALGO | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Exalgo Market Analysis and Sales Projections
What is Exalgo?
Exalgo (hydromorphone hydrochloride) is a prescription opioid analgesic indicated for the management of severe pain requiring daily, around-the-clock, long-term opioid treatment. It was approved by the FDA in 2010 and is marketed by Mallinckrodt Pharmaceuticals.
Market Overview
Therapeutic Area and Competition
Exalgo competes primarily within the opioid analgesics market, including drugs such as:
- Morphine ER
- Oxycodone ER
- Hydromorphone IR and ER formulations
- Fentanyl patches
- Methadone
The global opioid analgesics market was valued at approximately $9.4 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of around 3% until 2028. Growth drivers include increasing prevalence of chronic pain, aging populations, and regulatory approval pathways for new formulations.
Market Challenges
Key challenges to Exalgo include:
- Rising regulatory scrutiny over opioid use and abuse
- Increased adoption of non-opioid pain management options
- Litigation risks related to opioid epidemic
Regulatory and Legal Environment
The FDA has implemented REMS (Risk Evaluation and Mitigation Strategies) for opioids, affecting prescription patterns. US laws restrict prescribing and dispensing, impacting sales volume.
Current Market Position
Exalgo's market penetration is limited relative to other ER opioids. One reason is the availability of generic hydromorphone products, which have eroded the market share of branded formulations like Exalgo.
In the US, Exalgo's prescription volume has declined since its launch. Industry data indicates annual prescriptions peaked at approximately 200,000 units in 2013 but decreased to under 100,000 units by 2022 (IQVIA, 2023).
Sales Data (U.S.)
| Year | Estimated Sales | Units Prescribed | Notes |
|---|---|---|---|
| 2012 | $56 million | 200,000 units | Launch year |
| 2015 | $30 million | 120,000 units | Market consolidation |
| 2018 | $15 million | 80,000 units | Increased generic competition |
| 2022 | $10 million | 45,000 units | Continued decline |
Regional Market
Exalgo is mainly marketed in the US. International sales are limited due to regulatory uncertainties and market preferences.
Sales Projections
Assumptions
- Regulatory landscape remains restrictive with continued emphasis on opioid abuse mitigation.
- No significant new formulations or indications approved.
- Generic hydromorphone remains dominant for severe pain management.
Projection Scenarios
Conservative Scenario (low growth):
Sales decline 5% annually over five years, reaching approximately $5 million by 2028.
Moderate Scenario (stabilization):
Sales plateau at around $8 million for the next three years, then decline slowly due to market saturation and generics.
Aggressive Scenario (resurgence):
Introduction of abuse-deterrent formulations or expanded indications results in a 2% annual increase, reaching around $12 million by 2028.
| Year | Conservative ($ millions) | Moderate ($ millions) | Aggressive ($ millions) |
|---|---|---|---|
| 2023 | $9.5 | $9 | $10 |
| 2025 | $9 | $8.7 | $11.5 |
| 2028 | $5 | $8 | $12 |
Market Share Projection
Based on current trends, Exalgo's US market share within prescription opioids is projected to decline from approximately 0.6% in 2022 to below 0.4% by 2028.
Key Factors Impacting Future Sales
- FDA and CDC policy changes restricting opioid prescriptions.
- Competitor innovations, including abuse-deterrent formulations.
- Patent expirations and generic entry.
- Increased focus on alternative non-opioid therapies, such as nerve blocks and neuromodulation.
- Litigation and public health initiatives targeting opioid abuse.
Conclusion
Exalgo's sales are expected to decline over the next five years under most scenarios, constrained by regulatory and market dynamics. Moderate growth hinges on new formulations or expanded indications; absent this, decline is likely.
Key Takeaways
- Exalgo faces a shrinking market amid increasing regulatory restrictions and generic competition.
- US prescription volume peaked in 2013 and has seen a steady decline.
- Sales projections range from a continued decline to modest stabilization, with potential upsides from formulation innovations.
- Market share remains low and is likely to decline further.
- External factors, such as policy shifts and public health regulations, will heavily influence future sales.
FAQs
1. Why is Exalgo's market share declining?
Due to the availability of generic hydromorphone and heightened restrictions on opioid prescriptions, Exalgo's market share has decreased since launch.
2. What factors could reverse sales decline?
Introduction of abuse-deterrent formulations, establishing new indications, or regulatory changes favoring long-term opioid use could improve sales.
3. How does regulatory scrutiny impact Exalgo sales?
Stricter prescribing practices and enforcement of REMS policies limit access, reducing sales volume.
4. Are there opportunities in international markets?
Limited due to regulatory hurdles and differing pain management practices; however, emerging markets with increasing chronic pain may present future opportunities.
5. What KOLs influence Exalgo’s market?
Pain management specialists and regulatory authorities shape prescribing patterns through guidelines favoring or restricting opioid use.
Sources
[1] IQVIA. (2023). Opioid Prescription Trends.
[2] FDA. (2010). Exalgo (hydromorphone hydrochloride) approval letter.
[3] MarketWatch. (2022). Global Opioid Analgesics Market Size & Growth Forecast.
More… ↓
