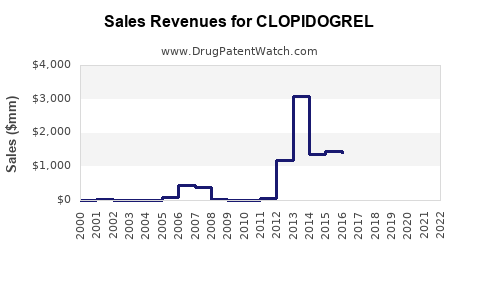
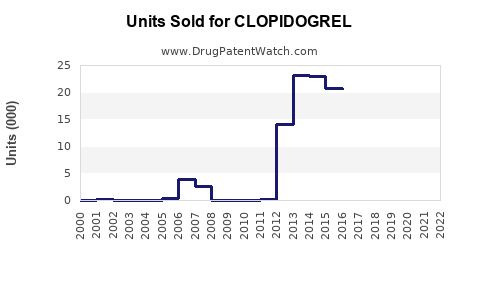
Last updated: February 20, 2026
Clopidogrel is an antiplatelet medication primarily indicated for preventing blood clots in patients with conditions such as acute coronary syndrome (ACS), peripheral artery disease, and ischemic stroke. The drug's widespread use and patent expirations influence market dynamics significantly.
Market Overview
The global clopidogrel market is driven by increasing prevalence of cardiovascular diseases (CVD), expanding aging populations, and rising awareness of antiplatelet therapy benefits. The drug's brand and generic segments exhibit different growth patterns.
Market Size (2022)
| Segment |
Value (USD billion) |
Growth Rate (CAGR 2022-2027) |
| Market Value |
5.2 |
4.8% |
| Brand Sales |
1.5 |
N/A |
| Generic Sales |
3.7 |
N/A |
The market was valued at approximately USD 5.2 billion in 2022, with generics dominating sales.
Key Players
- Pfizer (original developer, now other companies hold patents or manufactured generics)
- Teva Pharmaceutical Industries
- Mylan (now part of Viatris)
- Sun Pharmaceutical
- Aurobindo Pharma
Patent and Regulatory Status
Patent expiration impacts the market share, with Pfizer's patent expiring in many jurisdictions by 2012. Current sales are mostly from generics, leading to price competition.
Market Drivers
- Rising prevalence of coronary artery disease (CAD) and stroke globally.
- Increased screening and early intervention.
- Expanding use in combination therapy with other antithrombotic agents.
Market Challenges
- Competition from alternative therapies such as ticagrelor and prasugrel.
- Stringent regulatory environments.
- Concerns over bleeding risks impacting prescription patterns.
Sales Projections (2023-2028)
| Year |
Estimated Market Value (USD billion) |
Key Factors Influencing Growth |
| 2023 |
5.1 |
Stabilization after patent expirations; price decline in generics |
| 2024 |
5.3 |
Rising cardiovascular disease prevalence, new indications |
| 2025 |
5.6 |
Increased use in elderly and high-risk populations |
| 2026 |
6.0 |
Adoption of combination therapy, biosimilar entries |
| 2027 |
6.2 |
Continued growth, competitive pricing pressures |
| 2028 |
6.5 |
Market maturation, increased global penetration |
Compound annual growth rate (CAGR) averaging 4.8% from 2022-2027 predicts annual sales reaching approximately USD 6.5 billion by 2028.
regional Market Breakdown (2022)
| Region |
Market Share |
Growth Drivers |
| North America |
45% |
High CVD prevalence, well-established healthcare infrastructure |
| Europe |
25% |
Aging populations, regulatory approval of generics |
| Asia-Pacific |
20% |
Rising CVD incidence, expanding healthcare access |
| Other Regions |
10% |
Emerging markets with increasing diagnostic rates |
Strategic Considerations
- Patent cliffs and limited exclusivity encourage the proliferation of generics.
- Developing combination therapies and new formulations may recover some market share.
- Entry of biosimilars and biobetters presents future competition.
Key Takeaways
- The global clopidogrel market is valued at USD 5.2 billion in 2022, with generics accounting for the majority.
- Market growth is driven by rising CVD prevalence, demographic shifts, and expanded indications.
- Patents largely expired, leading to fierce price competition among generics.
- Sales are projected to increase to USD 6.5 billion by 2028, with a CAGR of 4.8%.
- Regional disparities exist, with North America maintaining dominant share due to high CVD rates and healthcare infrastructure.
FAQs
-
What factors influence the decline in brand sales for clopidogrel?
Patent expirations and the entry of generics reduce brand-level revenues, with price competition and formulary shifts favoring cheaper alternatives.
-
How does the emergence of newer antiplatelet agents impact clopidogrel sales?
Agents like ticagrelor and prasugrel, with reversible binding and different dosing, challenge clopidogrel's market share, especially in acute settings.
-
Are there geographic regions where clopidogrel is growing faster?
Asia-Pacific exhibits higher growth rates due to increasing CVD burden and expanding healthcare access.
-
What is the potential of biosimilars or biobetters for clopidogrel?
As patent protections end and patents of downstream formulations expire, biosimilar versions could enter the market, influencing prices and sales.
-
What regulatory issues could impact future sales?
Safety concerns, particularly regarding bleeding risks, and evolving guidelines on antithrombotic therapy may influence prescribing patterns.
References
[1] MarketResearch.com. (2023). Global Clopidogrel Market Analysis and Forecast.
[2] IQVIA. (2022). Pharmaceutical Market Reports.
[3] European Medicines Agency. (2022). Clopidogrel Summary of Product Characteristics.
[4] U.S. Food & Drug Administration. (2022). Drug Approvals and Labeling.
[5] World Health Organization. (2022). Cardiovascular Diseases Fact Sheet.