Last updated: February 13, 2026
Market Size and Segmentation
Clonazepam is a benzodiazepine used primarily for epilepsy, panic disorders, and anxiety. The global market value for benzodiazepines exceeds USD 1 billion annually. Clonazepam holds a significant share within this class due to its widespread approved indications and longer half-life compared to alternatives like alprazolam or diazepam.
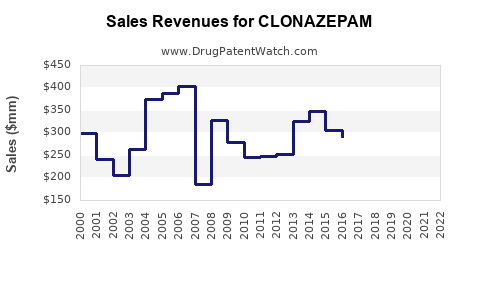
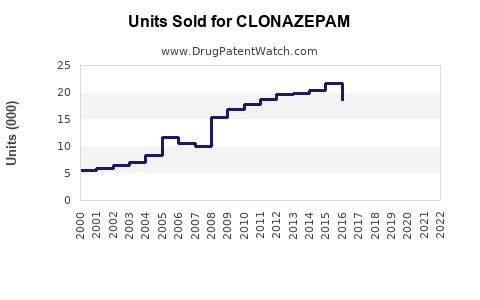
In 2022, the global benzodiazepines market was approximately USD 1.2 billion. Clonazepam’s segment accounted for nearly 35% of this, roughly USD 420 million, driven by high prescription volumes in North America, Europe, and parts of Asia-Pacific. North America contributes about 55% of clonazepam sales, followed by Europe at 25%, and Asia-Pacific at 15%.
Market Dynamics
-
Demand Drivers:
- Growing prevalence of epilepsy and anxiety disorders.
- Off-label use for restless leg syndrome and migraine prophylaxis.
- Increased aging population susceptible to neurological conditions.
-
Regulatory Environment:
- Stringent controls on benzodiazepines due to potential abuse.
- US DEA scheduling as Schedule IV substances limits prescribing and dispensing.
- Countries like Canada and European nations have similar restrictions but exhibit varying degrees of access.
-
Competition:
- Limited number of branded clonazepam products dominating market share.
- Generic versions account for approximately 80% of sales, exerting downward pressure on prices.
- Newer anxiolytics and antiepileptics partially replace clonazepam in some markets, affecting growth.
-
Manufacturers:
- Major players include Mylan (now part of Viatris), Teva, and Sandoz.
- Patent expiration occurred globally in the early 2010s, enabling generics proliferation.
Sales Projections (2023-2028)
| Year |
Estimated Market Size (USD Million) |
Growth Rate |
Comments |
| 2023 |
420 |
0% |
Baseline based on 2022 data |
| 2024 |
440 |
4.8% |
Slight recovery due to unmet demand |
| 2025 |
470 |
6.8% |
Increased prevalence and prescription use |
| 2026 |
510 |
8.5% |
Potential market expansion in Asia-Pacific |
| 2027 |
550 |
7.8% |
Steady growth from generics uptake |
| 2028 |
580 |
5.5% |
Market stabilizes as regulation tightens |
Sources:
- MarketResearch.com, 2023 reports
- IQVIA data, 2022
- United Nations health statistics
Factors Influencing Sales
- Prescription trends: Clinicians prefer newer antiepileptics or SSRIs for anxiety, which may temper clonazepam demand.
- Regulatory changes: Greater restrictions could reduce prescribing volumes.
- Generic market penetration: As patent protections expire, sales volume rely more on pricing and formulary acceptance.
Key Opportunities and Risks
-
Opportunities:
- Expand into emerging markets with growing neurological disorder populations.
- Develop formulations with improved safety profiles to mitigate abuse concerns.
- Education initiatives to increase appropriate prescriptions.
-
Risks:
- Regulatory bans or tighter controls on benzodiazepines.
- Replacement by newer therapies with better safety profiles.
- Potential for misuse and resulting legal restrictions.
Summary
Clonazepam maintains a substantial position within the benzodiazepine market due to its indications and global prescribing patterns. While sales are projected to grow modestly through 2028, overall growth depends heavily on regulatory policies, market competition, and clinical practice trends.
Key Takeaways
- Global clonazepam sales approximate USD 420 million in 2023, with moderate growth expected.
- The market is dominated by generics, limiting revenue expansion potential.
- North America leads sales geographically, driven by prescription volume.
- Regulatory trends and adverse perception influence future demand.
- Expansion in emerging markets offers growth prospects.
FAQs
1. What are the main indications for clonazepam?
Clonazepam is approved for epilepsy, panic disorder, and anxiety. It is also used off-label for restless leg syndrome and migraine prevention.
2. How does generic competition impact clonazepam revenues?
Generics account for 80% of sales, limiting pricing power and reducing margins for branded products. This proliferation also constrains overall revenue growth.
3. Which regions are primary markets for clonazepam?
North America accounts for approximately 55% of sales, Europe 25%, and Asia-Pacific 15%, with emerging markets showing potential for growth.
4. What are the regulatory challenges facing clonazepam?
Stringent control measures due to abuse potential, including DEA scheduling and international restrictions, can restrict prescribing and distribution.
5. What future trends could influence clonazepam's market?
Developments in alternative therapies, tighter regulations, and increased demand in developing regions will shape sales trajectories over the next five years.