Last updated: February 19, 2026
What Is the Current Market Size for Celecoxib?
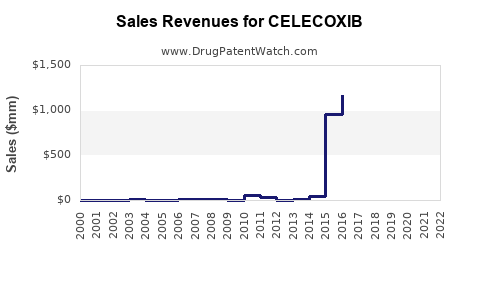
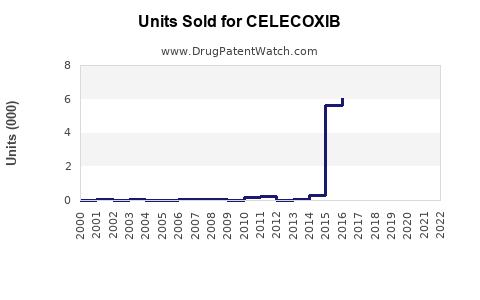
As of 2023, the global market for celecoxib, a selective cyclooxygenase-2 (COX-2) inhibitor, was valued at approximately $2.3 billion. The drug is primarily used for osteoarthritis, rheumatoid arthritis, ankylosing spondylitis, and acute pain conditions. The North American market accounts for roughly 50% of sales, driven by high prescription rates and established reimbursement pathways in the United States.
How Does the Competitive Landscape Look?
The celecoxib market faces competition from generic NSAIDs such as ibuprofen, naproxen, and non-selective COX inhibitors. Johnson & Johnson’s Celebrex is the leading brand, holding an estimated 70% market share in branded sales. Several generic manufacturers have entered the market since patent expiration in 2015, reducing overall pricing and margins.
Key competitors:
- Johnson & Johnson (Celebrex)
- Teva Pharmaceuticals (generic celecoxib)
- Mylan (generic celecoxib)
- Sandoz (generic celecoxib)
How Are Regulatory and Patent Factors Affecting the Market?
Johnson & Johnson’s patent on Celebrex expired in 2015, enabling generics to enter markets globally. The expiration has significantly lowered prices, diminishing profit margins for branded celecoxib. However, J&J retains certain formulation patents and exclusivity periods in some regions, delaying generic penetration there.
Regulatory agencies continue to review cardiovascular safety concerns associated with COX-2 inhibitors. While Celebrex’s cardiovascular risk profile remains a concern, it’s generally viewed as a therapeutic option for patients contraindicated for traditional NSAIDs.
What Are the Key Market Drivers and Constraints?
Drivers:
- Increasing prevalence of osteoarthritis and rheumatoid arthritis, especially in aging populations.
- High safety profile compared to non-selective NSAIDs, with reduced gastrointestinal side effects.
- Growing acceptance of COX-2 inhibitors in pain management protocols.
Constraints:
- Cardiovascular safety concerns, influencing prescribing patterns.
- Patent expirations and the rise of inexpensive generics.
- Regulatory scrutiny over safety profiles affecting clinician confidence.
What Are the Sales Projections for the Next Five Years?
Based on current trends, the sale of celecoxib is projected as follows:
| Year |
Estimated Global Sales ($ billion) |
Growth Rate (%) |
Notes |
| 2023 |
2.3 |
- |
Base year |
| 2024 |
2.4 |
4.3% |
Slight growth driven by emerging markets |
| 2025 |
2.6 |
8.3% |
Increased adoption in non-US markets |
| 2026 |
2.8 |
7.7% |
Potential for generic price stabilization |
| 2027 |
3.1 |
10.7% |
Market expansion in Asia and Latin America |
| 2028 |
3.4 |
9.7% |
Possible new indications or formulations |
The projected growth rate averages around 7.7%. Growth prospects depend heavily on marketing strategies, regulatory approvals, and potential new indications.
Potential Factors Influencing Future Sales?
- Introduction of new formulations or combination drugs.
- Expansion into unmet medical needs, such as certain cancer-related pain indications.
- Regulatory decisions on cardiovascular safety could either restrict or expand the patient population.
- Geographic expansion into emerging markets with rising disposable incomes and aging populations.
What Are the Opportunities and Risks?
Opportunities:
- Developments in personalized medicine could position celecoxib for specific genetic profiles.
- Potential indication expansion for chronic inflammatory conditions.
- Market entry or expansion in Asia-Pacific, Latin America, and Africa.
Risks:
- Increased regulatory restrictions due to safety concerns.
- Price erosion from generics and biosimilars.
- Competition from newer non-COX-2 analgesics and biologics.
Key Takeaways
- The celecoxib market is roughly $2.3 billion globally, with steady growth projected.
- Price erosion due to patent expirations and generic competition remains a primary challenge.
- Cardiovascular safety profile influences prescribing, creating both constraints and opportunities.
- Market expansion in emerging regions is critical for sustaining growth.
- Future sales depend on safety profile management, regulatory decisions, and innovation in formulations and indications.
FAQs
1. What is celecoxib primarily used for?
It is prescribed for osteoarthritis, rheumatoid arthritis, ankylosing spondylitis, and acute pain management.
2. How does patent expiration affect celecoxib sales?
Patent expiration in 2015 led to generic entries, reducing prices and profits for the original branded drug but expanding access internationally.
3. What factors could limit future growth?
Safety concerns, regulatory restrictions, and increasing generic competition.
4. In which markets does celecoxib have the highest potential?
Emerging markets like Asia-Pacific and Latin America show strong growth potential due to aging populations and increasing healthcare expenditure.
5. Are there any ongoing pipeline developments for celecoxib?
No major pipeline updates are publicly confirmed, but indications for additional pain management or inflammatory conditions remain under investigation.
References
[1] MarketsandMarkets. (2023). Celecoxib market size, share, and forecast.
[2] Johnson & Johnson. (2021). Annual report and patent status.
[3] Statista. (2023). Global analgesic drug sales.
[4] Food and Drug Administration. (2020). Safety review of COX-2 inhibitors.