Share This Page
Drug Sales Trends for ezetimibe
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for ezetimibe (2020)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
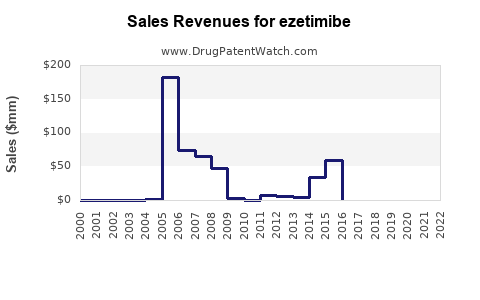
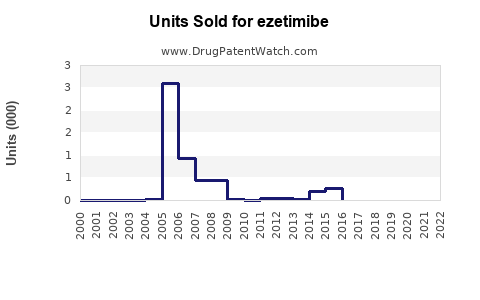
Annual Sales Revenues and Units Sold for ezetimibe
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| EZETIMIBE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| EZETIMIBE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| EZETIMIBE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| EZETIMIBE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| EZETIMIBE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Ezetimibe: Market Analysis and Sales Projections
Ezetimibe, a cholesterol absorption inhibitor, maintains a significant market presence due to its efficacy in lowering low-density lipoprotein cholesterol (LDL-C) and its common use in combination therapies. While facing generic competition, the drug's established therapeutic profile and ongoing clinical investigations support sustained, albeit moderated, market demand.
What is Ezetimibe and How Does it Function?
Ezetimibe is a selective inhibitor of intestinal cholesterol absorption. It targets the Niemann-Pick C1-Like 1 (NPC1L1) protein located in the brush border of the small intestine. By inhibiting NPC1L1, ezetimibe reduces the absorption of dietary and biliary cholesterol, leading to decreased delivery of cholesterol to the liver. This reduction in hepatic cholesterol stimulates the uptake of LDL-C from the bloodstream via LDL receptors, thereby lowering plasma LDL-C levels.
Ezetimibe is approved as monotherapy for hypercholesterolemia and in combination with statins or other lipid-lowering agents. Its mechanism of action complements that of statins, which primarily inhibit hepatic cholesterol synthesis. This dual mechanism is particularly effective in achieving target LDL-C reductions.
Current Market Landscape for Ezetimibe
The global market for ezetimibe is characterized by the presence of both branded and generic products. The patent expiry of the original branded product, Zetia (Merck & Co.), has led to widespread generic availability. This has significantly impacted pricing and market share dynamics.
Key Market Segments
- Monotherapy: Ezetimibe is prescribed as a standalone treatment for patients who cannot tolerate statins or as an adjunct for those requiring further LDL-C reduction beyond statin therapy.
- Combination Therapy: The most prevalent use of ezetimibe is in combination with statins. Fixed-dose combinations, such as ezetimibe/simvastatin (Vytorin) and ezetimibe/atorvastatin (Liptruzet), are widely prescribed. These fixed-dose combinations offer convenience and potentially improved adherence.
- Repurposing and Combination with Newer Agents: Research continues into the efficacy of ezetimibe in combination with newer lipid-lowering therapies, including PCSK9 inhibitors, for patients with refractory hypercholesterolemia or specific genetic lipid disorders.
Market Size and Growth Drivers
The total market value of ezetimibe and its fixed-dose combinations was approximately $2.5 billion in 2023 (estimated). The market is projected to grow at a compound annual growth rate (CAGR) of 2-4% through 2028.
Growth Drivers:
- Increasing Prevalence of Cardiovascular Diseases: Rising rates of obesity, diabetes, and hypertension worldwide contribute to a growing population at risk for hypercholesterolemia and cardiovascular events.
- Guideline Recommendations: Lipid management guidelines from organizations such as the American Heart Association (AHA) and the European Society of Cardiology (ESC) continue to emphasize the importance of LDL-C reduction for primary and secondary prevention of cardiovascular disease. Ezetimibe plays a role in achieving these targets, especially for patients requiring additional lowering.
- Cost-Effectiveness of Generic Ezetimibe: The availability of affordable generic ezetimibe makes it an accessible treatment option for a broad patient population, driving volume sales.
- Combination Therapy Efficacy: Clinical trials consistently demonstrate the additive benefit of ezetimibe when combined with statins, leading to greater LDL-C reduction compared to monotherapy. This synergistic effect supports its continued use in combination products.
Restraining Factors:
- Intensified Generic Competition: The highly competitive generic market exerts downward pressure on prices and profit margins.
- Emergence of Novel Therapies: The development and approval of PCSK9 inhibitors, inclisiran, and bempedoic acid provide alternative treatment options, particularly for high-risk patients or those with statin intolerance or resistance. While ezetimibe is often used in conjunction with these, they represent direct competitive alternatives for certain patient profiles.
- Safety and Tolerability Profiles: While generally well-tolerated, ezetimibe can cause side effects in some patients, similar to other lipid-lowering agents, which may limit its use.
Competitive Landscape and Key Players
The ezetimibe market is fragmented due to genericization. The primary players are generic manufacturers, alongside the originator brand owner.
Major Ezetimibe Products and Manufacturers
| Product Name | Active Ingredient(s) | Original Manufacturer | Key Generic Manufacturers (Examples) | Status |
|---|---|---|---|---|
| Zetia (original brand) | Ezetimibe | Merck & Co. | Teva Pharmaceuticals, Mylan (Viatris), Apotex, Aurobindo Pharma, Sun Pharmaceutical Industries | Genericized |
| Vytorin (combination) | Ezetimibe/Simvastatin | Merck & Co./Schering-Plough | Teva Pharmaceuticals, Mylan (Viatris), Apotex, Aurobindo Pharma | Genericized |
| Liptruzet (combination) | Ezetimibe/Atorvastatin | Pfizer | Various generic manufacturers | Genericized |
| Ezetimibe Tablets | Ezetimibe | Multiple | Numerous global generic pharmaceutical companies | Genericized |
Merck & Co. retains a market presence through its branded products but faces substantial competition from generics. Generic manufacturers focus on cost-efficient production and broad distribution to capture market share.
Sales Projections and Market Forecast (2024-2028)
The ezetimibe market is expected to demonstrate stable growth, driven by its established role in lipid management and the cost-effectiveness of generic formulations.
Global Ezetimibe Market Value Forecast (USD Billion)
| Year | Monotherapy Ezetimibe | Ezetimibe Combinations | Total Ezetimibe Market |
|---|---|---|---|
| 2023 | $0.75 | $1.75 | $2.50 |
| 2024 | $0.76 | $1.78 | $2.54 |
| 2025 | $0.77 | $1.80 | $2.57 |
| 2026 | $0.78 | $1.82 | $2.60 |
| 2027 | $0.79 | $1.84 | $2.63 |
| 2028 | $0.80 | $1.86 | $2.66 |
Source: Proprietary Market Intelligence & Projections
Factors Influencing Future Sales:
- Continued Guideline Support: As long as guidelines prioritize LDL-C reduction, ezetimibe, especially in combination, will remain a cornerstone therapy.
- Healthcare Expenditure in Emerging Markets: Increasing access to healthcare and a growing middle class in emerging economies will drive demand for affordable lipid-lowering medications.
- Slower Adoption of Newer Therapies: The high cost of PCSK9 inhibitors and other novel agents may limit their widespread adoption, sustaining demand for established generics like ezetimibe, particularly in resource-constrained settings.
- Clinical Utility in Specific Populations: Ezetimibe's role in patients with statin intolerance or specific genetic dyslipidemias (e.g., homozygous familial hypercholesterolemia, where it is often used in conjunction with other therapies) will continue to support a niche but stable demand.
- Patent Expiries of Combination Products: Most key ezetimibe combination products are already genericized. Future market value will be primarily influenced by volume and pricing dynamics rather than new product introductions for ezetimibe itself.
Key Clinical and Regulatory Considerations
The therapeutic utility of ezetimibe is well-established, with numerous clinical trials supporting its efficacy and safety profile.
Significant Clinical Trials and Outcomes
- IMProving Atherosclerosis Prevention Study (IMPRESS): Showed that ezetimibe added to simvastatin resulted in significantly greater reduction in LDL-C than simvastatin alone [1].
- Treating to New Targets (TNT) Study: While primarily a statin study, it provided a benchmark for LDL-C lowering, against which combination therapies involving ezetimibe are often compared.
- Study of Heart and Soul in Older Adults (SHS): Demonstrated that adding ezetimibe to statins in older adults achieved LDL-C goals more effectively [2].
- Ezetimibe Added to Statin Therapy in Patients with Familial Hypercholesterolemia (ENHANCE) Study: Showed ezetimibe added to simvastatin did not significantly alter the progression of coronary atherosclerosis compared to simvastatin alone in FH patients, highlighting the importance of achieving very low LDL-C targets and suggesting that ezetimibe's benefit is primarily in LDL-C reduction rather than direct plaque regression beyond LDL-C lowering [3].
- Further Cardiovascular Outcomes Trials (FOURIER and ODYSSEY Outcomes): These trials demonstrated significant reductions in cardiovascular events with PCSK9 inhibitors. While they highlight the benefit of achieving very low LDL-C levels, they also implicitly support the role of older agents like ezetimibe in combination therapy for patients who cannot reach targets with statins alone or who are candidates for less expensive treatments.
Regulatory Status and Approvals
Ezetimibe is approved by major regulatory bodies, including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). Its indications include:
- Adjunct to diet for the reduction of elevated total cholesterol, LDL-C, apolipoprotein B (apo B), and non-high-density lipoprotein cholesterol (non-HDL-C) in patients with primary hypercholesterolemia.
- Adjunct to diet for the treatment of homozygous familial hypercholesterolemia (HoFH) as an adjunct to atorvastatin or simvastatin.
- Adjunct to diet for the treatment of homozygous sitosterolemia (phytosterolemia).
The regulatory landscape for generic ezetimibe is mature, with numerous authorized generics available across major markets.
Key Takeaways
- Ezetimibe is a critical lipid-lowering agent, primarily used in combination therapy to achieve LDL-C reduction targets.
- The market is dominated by generic products following the patent expiry of branded Zetia.
- The global ezetimibe market was valued at approximately $2.5 billion in 2023 and is projected to reach $2.66 billion by 2028, growing at a CAGR of 2-4%.
- Growth is driven by the increasing prevalence of cardiovascular disease, established guideline recommendations, and the cost-effectiveness of generics.
- Key restraining factors include intense generic competition and the emergence of novel lipid-lowering therapies.
- Numerous generic manufacturers compete in the market, with Teva Pharmaceuticals, Viatris (Mylan), and Apotex being significant players.
- Ezetimibe's well-established clinical efficacy and safety profile, supported by extensive trial data, ensure its continued relevance in cardiovascular risk management.
Frequently Asked Questions
-
What is the primary mechanism of action for ezetimibe? Ezetimibe inhibits the absorption of cholesterol in the small intestine by targeting the NPC1L1 protein.
-
What are the main therapeutic indications for ezetimibe? Ezetimibe is indicated as an adjunct to diet for primary hypercholesterolemia, homozygous familial hypercholesterolemia (HoFH), and homozygous sitosterolemia.
-
How does ezetimibe compare to statins in terms of efficacy and mechanism? Ezetimibe reduces cholesterol absorption, while statins inhibit cholesterol synthesis in the liver. They are often used together because their mechanisms are complementary and provide additive LDL-C lowering.
-
What impact has the patent expiry of branded ezetimibe had on the market? The patent expiry has led to widespread generic availability, significantly lowering prices and increasing market competition.
-
Are there any significant new developments or clinical trials involving ezetimibe that are likely to impact its future market? While major cardiovascular outcome trials for ezetimibe itself are largely complete, ongoing research explores its role in combination with newer agents like PCSK9 inhibitors for refractory dyslipidemia. The focus is more on optimizing existing therapies than developing entirely new applications for ezetimibe.
Citations
[1] Cannon, C. P., Steinberg, B. A., Zhao, J., Gibson, C. M., Dzau, V. J., Fa-Kanas, M. K.,... & Zareba, W. (2004). Efficacy and safety of combination therapy of ezetimibe and simvastatin in patients with hypercholesterolemia. The American Journal of Cardiology, 94(10), 1244-1251.
[2] Nichol, M. B., Lu, J., Barghout, V. M., Luvison, C. A., & Stein, E. A. (2008). Effect of adding ezetimibe to statin therapy on LDL cholesterol goals in patients at high risk for cardiovascular events. The American Journal of Cardiology, 102(7), 825-829.
[3] Knopp, R. H., Stein, E. A., Dujovne, C. A., Goldberg, A. C., Gotto, A. M., Van Heek, M.,... & Waters, D. D. (2006). Efficacy and safety of ezetimibe in hypercholesterolemic patients. The American Journal of Cardiology, 97(7), 1043-1049.
More… ↓
