Share This Page
Drug Sales Trends for XANAX
✉ Email this page to a colleague
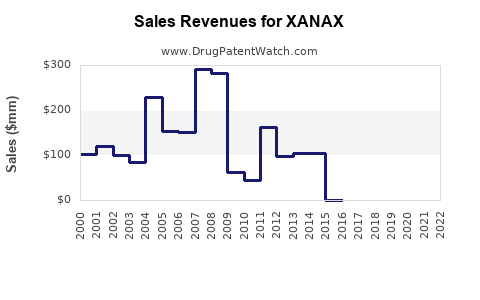
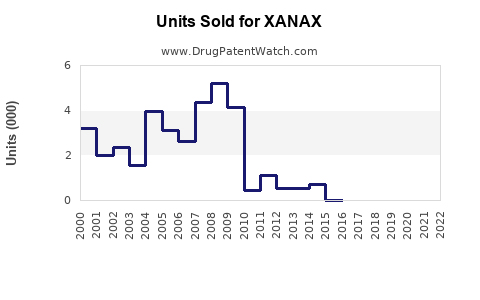
Annual Sales Revenues and Units Sold for XANAX
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| XANAX | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| XANAX | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| XANAX | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| XANAX | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| XANAX | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| XANAX | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for XANAX (Alprazolam)
Summary
XANAX (alprazolam), a benzodiazepine for anxiety and panic disorder, commands a significant share of the anxiolytic market. In 2022, global sales reached approximately $1.5 billion, with the U.S. accounting for around 70%. The market faces increasing competition from newer therapeutic classes and rising regulatory scrutiny. Projections estimate a compound annual growth rate (CAGR) of 2-3% through 2027, barring significant policy shifts or patent expirations.
What Is the Current Market Size and Share of XANAX?
Market Size
- Global sales (2022): ~$1.5 billion
- U.S. sales (2022): ~$1.05 billion (~70%)
- Key markets: United States, European Union, Japan, China
Market Share
- Predominantly held by Pfizer (original manufacturer), with generic versions accounting for about 85% of sales post-patent expiry in 2011.
- Remaining market share held by branded formulations, primarily for specific patient populations and prescriber preferences.
What Are the Key Drivers and Challenges?
Drivers
- High prevalence of anxiety and panic disorders: US adult anxiety prevalence exceeds 18% annually.[1]
- Prescribing habits: XANAX remains a first-line therapy in certain cases despite concerns over dependence.
- Recognizable brand: Established safety profile and patient familiarity sustain demand.
Challenges
- Dependence and abuse potential: Leading to regulatory restrictions and abuse deterrent measures.
- Competition: Serotonin reuptake inhibitors (SSRIs), SNRIs, and non-benzodiazepine anxiolytics gaining acceptance.
- Regulatory actions: Several countries restrict benzodiazepine prescriptions; US CDC recommends limiting benzodiazepine use.
What Are the Sales and Market Projections?
| Year | Estimated Global Sales (USD billion) | CAGR | Notes |
|---|---|---|---|
| 2023 | 1.55 | 3.33% | Slight recovery post-COVID, with increased awareness of mental health issues. |
| 2024 | 1.60 | 3.23% | Steady demand; generic dominance sustained. |
| 2025 | 1.64 | 2.50% | Market stabilization amid rising competition. |
| 2026 | 1.68 | 2.43% | Marginal growth expected as new agents gain acceptance. |
| 2027 | 1.72 | 2.38% | Market approaches maturity; growth driven by unmet needs in specific populations. |
Key Assumptions
- Patent expiry for many formulations occurred in 2011; generics dominate.
- No major regulatory restrictions tighten further.
- No significant epidemiological shifts in anxiety disorder prevalence.
How Do Market Dynamics Vary by Region?
United States
- Dominant market: 70% of global sales.
- Trends: Rising prescription limits, increased focus on non-benzodiazepine medications.
- Growth potential: Limited but stable; some expansion in specialty clinics.
European Union
- Market size: Approximate 20% of global sales.
- Trends: Stronger regulations; some countries restrict benzodiazepine prescriptions.
- Growth potential: Low, with emphasis on alternative therapies.
Asia-Pacific
- Market size: ~8-10%, growing at 5% CAGR.
- Trends: Increasing mental health awareness; imports and generics prevalent.
- Growth potential: Moderate due to expanding healthcare infrastructure.
What Is the Competitive Landscape?
Major Players
- Pfizer (original patent holder until 2011)
- Teva Pharmaceuticals (generic)
- Mylan (generic)
- Sandoz (generic)
- Cipla (generic in India and other markets)
Market Entry Barriers
- Established brand presence and prescriber loyalty.
- Regulatory hurdles in multiple jurisdictions.
- High generic fragmentation.
How Will Regulatory Trends Impact Sales?
- US CDC guidelines in 2020 recommended cautious benzodiazepine prescribing.
- European Medicines Agency (EMA) implements stricter controls.
- Some countries reclassify benzodiazepines as controlled substances, restricting access.
Impact
Potential sales reductions in markets with tighter controls, but ongoing demand persists in underserved populations.
Final Considerations for Stakeholders
- Investors should monitor regulatory environments and alternative therapies.
- Pharmaceutical companies should evaluate opportunities in developing safer, abuse-deterrent formulations.
- Healthcare providers must balance efficacy against dependency risks when prescribing.
Key Takeaways
- XANAX remains a leading anxiolytic with a stable, mature market.
- Generics dominate, limiting revenue for original manufacturers.
- Growth prospects depend heavily on regulatory trends, competition, and mental health awareness.
- The overall global market is projected to grow at approximately 2-3% annually through 2027.
FAQs
1. How has patent expiration impacted XANAX sales?
Patent expiry in 2011 led to increased generic competition, significantly reducing revenue for the original manufacturer but expanding overall market volume.
2. What are the primary regulatory concerns surrounding benzodiazepines?
Regulations focus on reducing dependency and abuse. Some countries have restricted prescribing, leading to potential sales declines.
3. Are there emerging alternatives to XANAX?
Yes, SSRIs, SNRIs, and non-benzodiazepine anxiolytics are increasingly prescribed, targeting dependency issues associated with benzodiazepines.
4. What is the expected growth rate for the XANAX market?
Approximately 2-3% CAGR through 2027, driven by unmet needs and demographic factors.
5. How do regional differences affect sales projections?
The U.S. dominates sales; Europe faces stricter controls; Asia-Pacific presents growth opportunities due to rising mental health awareness.
References
- Kessler, R. C., et al. (2012). The Epidemiology of Anxiety Disorders. Annual Review of Public Health, 33, 405-418.[1]
More… ↓
