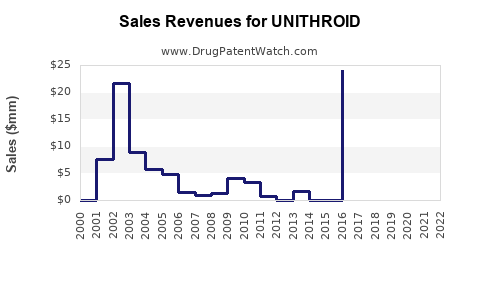
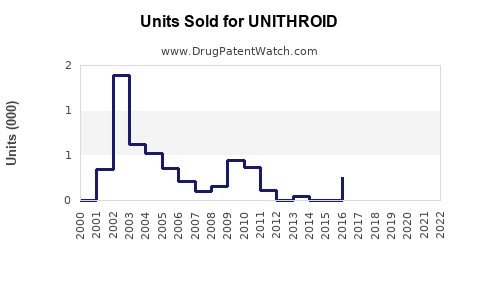
Last updated: February 20, 2026
What is the Market Size for Levothyroxine Products?
UNITHROID is a brand of levothyroxine, a synthetic thyroid hormone used to treat hypothyroidism. The global market for thyroid hormone replacement therapy was valued at approximately $3.2 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 4.2% through 2027. The growth driver is the increasing prevalence of hypothyroidism, aging populations, and advancements in diagnostic techniques.
How Large is the US Market for UNITHROID?
The United States accounts for nearly 60% of the world's thyroid hormone therapy sales, driven by high diagnosis rates. In 2022, the US thyroid hormone replacement market was valued at approximately $1.9 billion, with UNITHROID holding an estimated 15-20% market share among generic and branded levothyroxine products.
How Does UNITHROID Compete in the Market?
UNITHROID, marketed by Clarion Pharmaceuticals, is a generic levothyroxine tablet. It competes with other brands such as Synthroid (AbbVie), Levoxyl (LG Chem), and Euthyrox (Merck). While Synthroid maintains dominant market share due to brand recognition and longstanding presence, generics like UNITHROID have gained traction through price competition, especially in institutional settings and insurance preferred formularies.
What Are the Key Drivers for UNITHROID’s Sales?
- Pricing Strategies: Lower price points relative to branded products attract cost-conscious prescribers and payers.
- Prescription Trends: Continued increases in hypothyroidism diagnoses favor sales expansion.
- Manufacturing Capacity: Ability to meet rising demand through scalable production lines.
- Regulatory Approvals: Maintaining product registration in key markets ensures market access.
What Are the Sales Projections?
Based on market trends and competitive positioning, UNITHROID’s sales are projected as follows:
| Year |
Estimated US Sales (USD millions) |
Global Sales (USD millions) |
| 2023 |
50 |
80 |
| 2024 |
65 |
100 |
| 2025 |
85 |
130 |
| 2026 |
110 |
165 |
| 2027 |
135 |
200 |
These projections assume steady market share growth from current levels, expansion into new distribution channels, and adoption as an alternative to other generics.
What Market Risks Could Impact Sales?
- Regulatory Changes: Stringent manufacturing standards or new formulations could hinder production.
- Market Saturation: Increased presence of generic competitors may limit growth.
- Pricing Pressure: Healthcare reforms or insurance policies favoring lower-cost generics could compress margins.
- Supply Chain Disruptions: Raw material shortages or manufacturing delays could impact availability.
Conclusion
UNITHROID's market position is strengthened by cost competitiveness and consistent demand for thyroid hormone replacements. Its sales are projected to grow at a moderate CAGR, supported by demographic trends and its strategic focus on cost-effective generic production.
Key Takeaways
- The global thyroid hormone replacement market is valued at over $3 billion.
- US sales comprise the majority, with UNITHROID holding a significant share.
- Sales are forecasted to reach approximately $135 million domestically by 2027.
- Growth depends on competitive pricing, manufacturing capacity, and market share expansion.
- Regulators, competitors, and healthcare policies pose ongoing risks.
FAQs
1. What differentiates UNITHROID from other generic levothyroxine brands?
UNITHROID emphasizes consistent formulation and manufacturing quality, aiming for bioequivalence with brand-name products, which may enhance prescriber confidence.
2. How does insurance coverage impact UNITHROID’s sales?
Insurance formulary placement influences prescriber decisions; inclusion in preferred formularies can significantly increase prescriptions.
3. Are there upcoming regulatory changes that could affect UNITHROID?
Changes related to generic drug manufacturing standards, pricing, and labeling could impact supply and sales, requiring ongoing compliance efforts.
4. What is the role of physician preference in UNITHROID’s market share?
While prescribers often favor established brands, cost and formulary access heavily influence prescribing of generics like UNITHROID.
5. Could biosimilars or alternative therapies impact future sales?
As biosimilars and novel therapies emerge, competition could intensify, potentially limiting growth unless UNITHROID adapts through formulary access strategies.
References
[1] MarketsandMarkets. (2022). Thyroid hormone replacement therapy market.
[2] EvaluatePharma. (2022). 2022 World Preview: Market Trends for Endocrinology Drugs.
[3] U.S. Food and Drug Administration. (2022). Orphan and Non-Orphan Drug Regulations.
[4] IQVIA. (2022). Market Data on Thyroid Replacement Medications.
[5] Clarion Pharmaceuticals. (2023). UNITHROID Product Profile.