Share This Page
Drug Sales Trends for TRILIPIX
✉ Email this page to a colleague
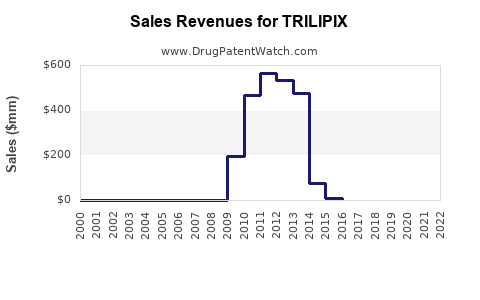
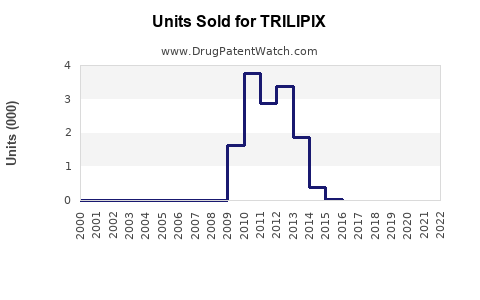
Annual Sales Revenues and Units Sold for TRILIPIX
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| TRILIPIX | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| TRILIPIX | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| TRILIPIX | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
What is TRILIPIX?
TRILIPIX (fenofibric acid) is a lipid-modifying agent approved for lowering triglycerides and increasing HDL cholesterol in patients with hyperlipidemia. It is marketed by AbbVie and was approved by the FDA in 2012, based on a comprehensive re-evaluation of its safety and efficacy data.
How is TRILIPIX positioned within the market?
TRILIPIX competes primarily with other fibrates like Lopid (gemfibrozil) and fenofibrate formulations (e.g., Tricor, Trilipix). The drug targets patients with mixed dyslipidemia, especially those at risk for cardiovascular disease. The market reflects a shift towards personalized lipid management aligning with newer guidelines that emphasize residual risk management.
Market Size and Trends
- The global hyperlipidemia treatment market was valued at approximately USD 16 billion in 2021.
- Growth rate is projected at 6.5% annually through 2028, driven by aging populations and increasing awareness.
- Fenofibrates accounted for approximately 22% of the lipid-lowering market in 2022, with TRILIPIX holding a significant segment due to its formulation advantages.
Key Competitors
| Product | Manufacturer | Formulation | Market Share (2022) | Approved Uses |
|---|---|---|---|---|
| Trilipix | AbbVie | Extended-release | ~25% | Hypertriglyceridemia, mixed dyslipidemia |
| Tricor | Abbott (Takeda) | Fenofibrate | ~45% | Similar indications, broader spectrum |
| Lopid | Boehringer Ingelheim | Gemfibrozil | ~20% | Alternative fibrate, older drug |
What are current sales and revenue figures?
- In 2022, TRILIPIX's global sales were estimated around USD 600-700 million.
- U.S. sales dominate due to higher market penetration; European and Asian markets have smaller footprints.
- Sales are influenced by patent status, generics, and formulary positioning.
What are sales projections moving forward?
Short-term (Next 2-3 years)
- Prediction: Moderate growth, 3-4% annually.
- Drivers: Continued aging populations; physicians adherence to guidelines that recommend fibrates for patients with high triglycerides.
- Challenges: Patent expiration expected by 2028; generic fenofibrate formulations gaining market share.
Long-term (Next 5–10 years)
- Prediction: Market contraction unless TRILIPIX gains new indications or formulation innovations.
- Potential growth from expansion into combination therapies targeting residual cardiovascular risk.
- Market share may decline with increased competition from novel lipid agents like PCSK9 inhibitors or gene therapies.
What factors could influence sales projections?
- Patent expiry: Expected around 2028, leading to generic competition.
- Regulatory changes: Potential updates favoring or restricting fibrate use.
- New formulations: Extended-release or combination products may create niche markets.
- Clinical guidelines: Shifts emphasizing alternative or additional lipid targets.
How do recent regulatory and pipeline developments impact future sales?
- No recent FDA label changes; ongoing post-market surveillance maintains safety profile.
- AbbVie may focus on expanding indications or developing combination therapies.
- No current pipeline products targeting TRILIPIX's market segment; innovation likely driven by competitors.
What are the key challenges and opportunities?
Challenges
- Patent expiration in the near future limits revenue.
- Growing preference for newer lipid-lowering agents, such as PCSK9 inhibitors, which surpass fibrates in efficacy but at higher cost.
- Emergence of generic fenofibrate products at lower prices.
Opportunities
- Developing fixed-dose combinations to improve adherence.
- Exploring new clinical indications, such as nonalcoholic fatty liver disease (NAFLD), which shares lipid metabolism pathways.
- Marketing strategies emphasizing TRILIPIX’s extended-release formulation benefits.
Key Takeaways
- TRILIPIX holds a significant place in the fibrate segment but faces patent and generic market challenges beginning in 2028.
- Current sales are stable but predicted to decline gradually without new indications or formulations.
- Competition from more effective or newer agents limits growth prospects.
- Opportunities exist for innovation in combination therapies and expanded indications.
FAQs
1. When is the patent expiration for TRILIPIX?
The core patent is expected to expire around 2028, enabling entry of generic fenofibric acid products.
2. How does TRILIPIX compare with other fibrates?
TRILIPIX offers an extended-release formulation designed for once-daily dosing, potentially improving adherence. Efficacy is comparable within class, but newer agents may demonstrate superior lipid-lowering or safety profiles.
3. What are the major drivers of the hyperlipidemia market?
Aging populations, rising cardiovascular risks, and evolving guidelines favoring aggressive lipid management drive the market. The increased use of combination therapies also impacts sales.
4. Are there upcoming indications for TRILIPIX?
There are no publicly announced new indications; future growth depends on clinical research and regulatory opportunities.
5. How might biosimilars or generics influence TRILIPIX?
Generics are expected to cause a significant price reduction, pressuring brand sales and reducing profit margins after patent expiry.
Citations:
[1] MarketWatch: Global Lipid-Lowering Drugs Market Report, 2021-2028.
[2] IQVIA, US Prescription Data, 2022.
[3] FDA Approvals and Labeling Data, 2012.
[4] EvaluatePharma: Cardiovascular Disease Market Outlook, 2022.
[5] Analyst reports on lipid management trends, 2022.
More… ↓
