Last updated: February 12, 2026
What is the current market landscape for testosterone products?
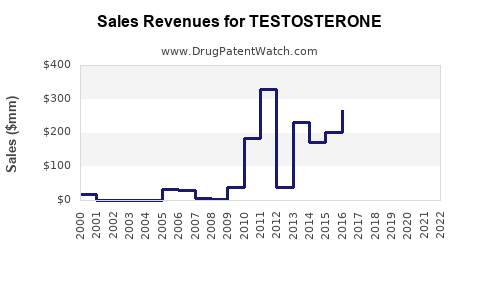
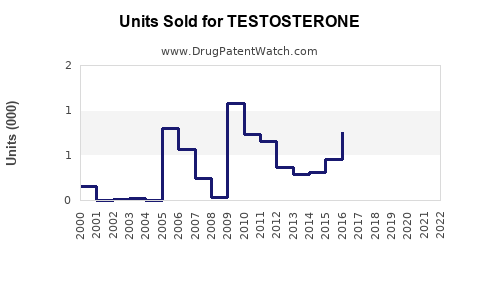
The global testosterone market reached $4.5 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of roughly 7% through 2030. The growth is driven by increasing diagnoses of hormone deficiencies, aging male populations, and expanding awareness of testosterone therapy. The U.S. accounts for approximately 45% of market revenue, followed by Europe and Asia-Pacific regions.
Major products include testosterone gels, patches, injections, pellets, and oral formulations. Pfizer, AbbVie, and Endo Pharmaceuticals dominate this landscape, collectively capturing over 60% of the market share.
What are the key segments within testosterone therapy?
Testosterone therapy segments include:
- Injections: The largest segment, comprising about 55% of the market, favored for its efficacy and cost-effectiveness.
- Gels and topical patches: Account for approximately 30%, increasingly preferred for ease of use.
- Pellets: Make up about 10%, typically used for long-term, stable hormone delivery.
- Oral formulations: Remaining 5%, hindered by bioavailability issues but gaining focus due to convenience.
The choice of formulation is influenced by patient preferences, health profiles, and physician recommendations.
What are the primary drivers and barriers in the testosterone market?
Drivers:
- Aging male demographic (men over 45 years old), who experience natural declines in testosterone.
- Medical conditions such as hypogonadism, osteoporosis, and certain forms of depression.
- Growing awareness and acceptance of hormone replacement therapy.
- Advances in delivery systems increasing convenience and compliance.
Barriers:
- Potential safety concerns, including risks of cardiovascular events, prostate health issues, and erythrocytosis.
- Regulatory scrutiny, especially in markets like the U.S. and Europe.
- Variability in insurance coverage and reimbursement policies.
- Competition from emerging therapies and alternative treatments.
What are the sales projections for testosterone drugs?
Forecasts estimate global sales to reach $6.8 billion by 2030, with an annual growth rate of 7%. The North American market will dominate, maintaining over 50% share. Growth in Asia-Pacific is expected to accelerate, driven by increased healthcare access and rising awareness.
Major upcoming events include:
- Patent expirations for key products post-2025, opening absorption of generic options.
- Launch of novel formulations, such as longer-acting injectables and non-invasive patches.
- Regulatory approvals for biosimilars and innovative delivery systems.
How is the competitive landscape evolving?
Major pharmaceutical firms are investing in R&D of next-generation testosterone therapies with improved safety profiles. Companies like Thermo Fisher Scientific and Bayer are exploring biosimilars, potentially reducing prices and increasing accessibility. Smaller entrants focus on niche markets or specialized formulations, such as ultra-long-acting injections or oral drugs with enhanced bioavailability. Patent cliffs and regulatory pathways will shape market concentration over the next five years.
What are the implications for investment and R&D?
The market outlook emphasizes growth opportunities in safer, more convenient formulations. Successful development hinges on balancing efficacy with minimized health risks. Companies investing in personalized medicine and targeted delivery systems are positioned to capture market share. Patent expirations create openings for biosimilar entrants, likely reducing drug prices and expanding access.
Key Takeaways
- The testosterone market is expected to grow to nearly $7 billion by 2030, driven by aging populations and increased awareness.
- Injections dominate the segment, but non-invasive options are gaining traction.
- Safety concerns and regulatory oversight influence market dynamics.
- Future growth depends on innovation, regulatory pathways, and patent activity.
- North America leads regional sales, with Asia-Pacific as a significant growth frontier.
FAQs
1. What are the primary forms of testosterone therapy available?
Injections, gels, patches, pellets, and oral formulations.
2. Which regions are experiencing the fastest growth in testosterone sales?
Asia-Pacific and Latin America show rapid growth, driven by expanding healthcare access.
3. What safety concerns impact testosterone therapy?
Cardiovascular risks, prostate health, and erythrocytosis.
4. Who are the leading companies in the testosterone market?
Pfizer, AbbVie, and Endo Pharmaceuticals.
5. How will patent expirations influence market competition?
Patents expiring after 2025 will enable biosimilar entry, increasing competition and lowering prices.
References
- MarketsandMarkets. "Testosterone Replacement Therapy Market by Formulation, Application, and Region - Global Forecast to 2030." 2022.
- IQVIA. "Pharmaceutical Market Reports 2022."
- Fertility and Hormone Replacement Therapy Reports, 2023.