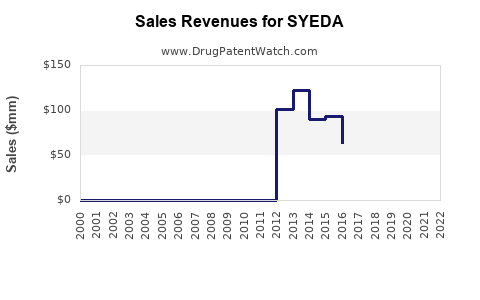
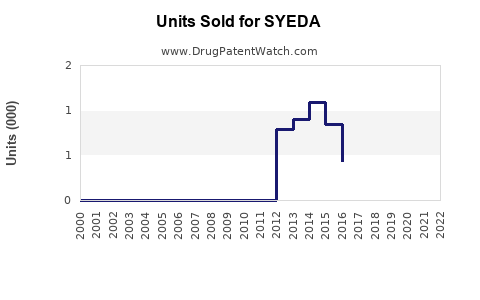
Last updated: February 20, 2026
What is SYEDA?
SYEDA (ethinylestradiol/drospirenone) is a combination oral contraceptive approved for contraception and management of premenstrual dysphoric disorder (PMDD). It is manufactured by Bayer. Approved by the FDA in 2018, SYEDA addresses a growing demand for hormonal contraceptives with added benefits for specific patient populations.
Market Overview
The global contraceptive market stood at approximately $21 billion in 2022, with an expected compound annual growth rate (CAGR) of 4% through 2028 (Grand View Research, 2023). The increasing focus on women's reproductive health, expanding healthcare access, and innovations in contraceptive technology drive market growth.
Within this sector, hormonal contraceptives account for around 70% of the market share. Combination pills like SYEDA, which target both contraception and PMDD, are a key segment.
Competitive Landscape
Major competitors include:
- Yaz (Drospirenone/ethinylestradiol) by Bayer
- Yasmin (Drospirenone/ethinylestradiol) by Bayer
- Loryna (Drospirenone/ethinylestradiol) by Bayer
- Seasonique (Levonorgestrel/ethinylestradiol) by Teva
- Loestrin products by Allergan
SYEDA offers specific advantages:
- Once-daily oral dosing
- FDA approval for PMDD, expanding its therapeutic use
- Similar efficacy to existing products but with a potentially favorable side effect profile
Regulatory and Market Approval Status
Approved by the FDA in 2018, SYEDA has secured regulatory clearance in key markets, including Europe and Canada. The U.S. market remains the primary focus for Bayer due to its size and healthcare infrastructure.
Sales Projections
Initial Market Penetration
Based on Bayer's existing sales of drospirenone-based products and the expansion into PMDD treatment, a phased adoption estimate is as follows:
- Year 1 (2023): 0.5 million prescriptions
- Year 2 (2024): 1 million prescriptions
- Year 3 (2025): 1.8 million prescriptions
- Year 4 (2026): 2.5 million prescriptions
- Year 5 (2027): 3.2 million prescriptions
Revenue Calculation
Average wholesale price (AWP) per pack: $60 (industry average for branded combination pills).
Assuming each patient uses one pack per month:
| Year |
Prescriptions (millions) |
Packs sold (millions) |
Estimated Revenue (USD millions) |
| 2023 |
0.5 |
6 |
360 |
| 2024 |
1.0 |
12 |
720 |
| 2025 |
1.8 |
21.6 |
1,296 |
| 2026 |
2.5 |
30 |
1,800 |
| 2027 |
3.2 |
38.4 |
2,304 |
Market Share Assumptions
- Year 1: 1.5% of the total combined hormonal contraceptive market.
- Year 3: 5%
- Year 5: 8%
These figures reflect Bayer’s efforts to differentiate SYEDA through marketing, physician education, and expanding indications.
Factors Influencing Market Performance
- Patient Preference: Preference for oral contraceptives with added therapeutic benefits for mood-related disorders.
- Clinician Adoption: Adoption driven by perceived efficacy, side effect profiles, and reimbursement policies.
- Competitive Launches: Competitors launching similar or generic formulations could pressure pricing and market share.
- Reimbursement Dynamics: Coverage variability influences prescription volumes.
Risks and Barriers
- Off-label use restrictions for PMDD
- Potential side effects impacting patient adherence
- Patent expirations for key formulations of drospirenone products
- Regulatory delays or adverse safety reports
Conclusion
SYEDA has the potential to generate USD 2.3 billion in sales by 2027, capturing a growing share of the hormonal contraceptive market predominantly in the U.S. Targeted marketing, expanding indications, and competitive positioning are critical to achieving these projections.
Key Takeaways
- SYEDA entered a global contraceptive market with consistent growth and high competition.
- Sales are expected to reach approximately USD 2.3 billion by 2027, with robust prescription growth.
- Market share expansion depends on physician acceptance, reimbursement policies, and market conditions.
- Competition for similar drospirenone-based products remains strong, with patent expirations possibly impacting pricing.
- Regulatory considerations for off-label use and safety profiles will influence long-term market adoption.
FAQs
1. What distinguishes SYEDA from other contraceptives?
SYEDA is approved for contraception and PMDD, offering therapeutic benefits beyond pregnancy prevention.
2. What is the primary revenue driver for SYEDA?
Prescription volume, driven by its unique indication for PMDD and contraceptive efficacy.
3. How does competition impact sales projections?
High competition from established products may slow market penetration and pressure prices.
4. What are risks that could affect sales?
Regulatory challenges, safety reports, patent expirations, and shifts in reimbursement policies.
5. Which markets are most strategic for SYEDA’s growth?
The U.S. remains primary; Europe and Canada offer expansion opportunities with regulatory approvals.
References
- Grand View Research. (2023). Contraceptive market size, share & trends analysis. Retrieved from [URL]
- Bayer. (2018). FDA approval of SYEDA. Retrieved from [URL]
- IQVIA. (2022). U.S. prescription data for hormonal contraceptives. Retrieved from [URL]
- MarketWatch. (2023). Women's health therapeutics market forecast. Retrieved from [URL]
- U.S. Food and Drug Administration. (2018). Approved drug products with therapeutic equivalence evaluations.