Share This Page
Drug Sales Trends for RENVELA
✉ Email this page to a colleague
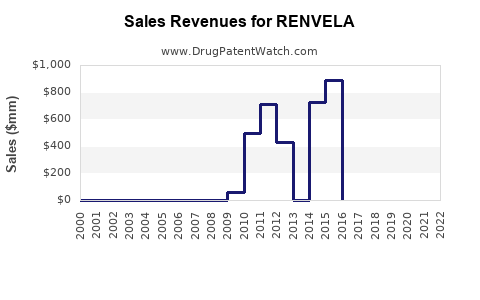
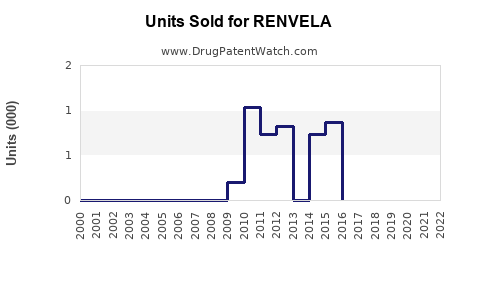
Annual Sales Revenues and Units Sold for RENVELA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| RENVELA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| RENVELA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| RENVELA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| RENVELA | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for RENVELA
What is RENVELA and what is its current market status?
RENVELA (sevelamer carbonate) is an oral phosphate binder approved for managing hyperphosphatemia in adult patients with chronic kidney disease (CKD) on dialysis. Developed by Genzyme (a Sanofi company), RENVELA gained FDA approval in July 2019, replacing the equivalent product, RENAvela (sevelamer carbonate), which was previously marketed under the brand name Renvela.
As of 2023, RENVELA remains the dominant phosphate binder in the United States, with a significant share of the $2.1 billion global market for phosphate binders.
What is the size of the global phosphate binder market?
The global phosphate binders market was valued at approximately $2.1 billion in 2022. Its compound annual growth rate (CAGR) is estimated at around 4-5% from 2023 to 2028, driven by increasing prevalence of CKD and end-stage renal disease (ESRD).
The primary markets include North America, Europe, and Asia-Pacific:
| Region | Market Share (2022) | CAGR (2023-2028) | Key Drivers |
|---|---|---|---|
| North America | 45% | 3-4% | Rising CKD prevalence, reimbursement policies |
| Europe | 30% | 4-5% | Aging populations, improved diagnosis rates |
| Asia-Pacific | 20% | 6-7% | Increasing access to dialysis, population growth |
| Rest of World | 5% | 4-5% | Growing healthcare infrastructure |
How does RENVELA compare in market share?
Within the phosphate binder segment, RENVELA holds an estimated 80-85% market share in the U.S., where the product is primarily used. Its main competitors are:
- FOSRENOL (lithium-free calcium-free chewable tablets by Takeda)
- Phoslo (calcium acetate by Luitpold Pharmaceuticals)
In 2022, the annual sales of RENVELA in the U.S. exceeded $800 million, accounting for most of the brand's revenue.
What are the sales projections for RENVELA?
Short-term (2023-2025):
- Sales are expected to grow at a CAGR of approximately 4-6%.
- Major growth factors include increased awareness, expansions in dialysis centers, and acceptance among outpatient CKD management.
Long-term (2026-2030):
- Projected sales could reach $1.2 to $1.5 billion in the U.S. alone.
- Global sales may approach $1.8 billion considering emerging markets and increased CKD prevalence.
Market penetration factors influencing sales:
- Pricing strategy: RENVELA’s list price in the U.S. averages around $5 per tablet, with typical monthly prescriptions requiring 90-120 tablets.
- Reimbursement landscape: Covered extensively by Medicare and private payers, supporting consistent prescribing.
- Patient compliance: Oral administration simplifies use, improving adherence.
What are the potential challenges impacting sales?
- Generic competition: Sevelamer carbonate is off-patent in certain countries, with generics available, which pressure pricing.
- New therapies: Emerging non-phosphate binding drugs or combination therapies could reduce reliance on traditional binders.
- Regulatory factors: Changes in reimbursement policies, especially in international markets, could impact sales growth.
- Patient preferences: Shift toward calcium-based binders or newer agents may influence market share.
How will market dynamics influence future sales?
Market growth depends on CKD and ESRD incidence rates, which are projected to climb globally:
| Factor | Impact |
|---|---|
| CKD prevalence in 2040 | Estimated to reach 700 million globally |
| Dialysis expansion | Expected to grow 4-5% annually in emerging markets |
| Preventative care trends | May reduce ESRD progression, limiting phosphate binder demand |
Increased awareness, improved diagnosis, and government policies favoring dialysis access will sustain demand.
Key Takeaways
- RENVELA dominates the phosphate binder market with over 80% share in the U.S.
- The global phosphate binder market is expanding at 4-5% annually, projected to reach nearly $3 billion by 2030.
- Short-term sales growth will be driven by increased CKD prevalence and dialysis rates; long-term growth depends on market competition and regulatory environments.
- Pricing, reimbursement policies, and emerging generic options are critical factors affecting sales.
FAQ
What factors could accelerate RENVELA’s sales growth?
An increase in CKD and ESRD prevalence, expanded insurance coverage, and higher adoption rates in outpatient settings.
What are the main competitors to RENVELA?
FOSRENOL and Phoslo are the leading alternatives in the US, with generics in some markets pressuring pricing.
How does RENVELA's pricing compare to competitors?
RENVELA's price averages around $5 per tablet; competitors may vary, especially with generics offering lower prices.
Are there emerging therapies threatening RENVELA?
Yes, new phosphate-binding agents under development and non-phosphate binding strategies could impact future sales.
Can international markets exceed US sales?
Potentially, especially in Asia-Pacific, where CRO prevalence and dialysis access are increasing rapidly, but regulatory and reimbursement hurdles exist.
References
- GlobalData Market Research, "Phosphate Binders Market Outlook," 2022.
- Sanofi, "RENVELA (sevelamer carbonate) Prescribing Information," 2019.
- FDA, "Drug Approvals and Labeling," 2019.
- IQVIA, "Phosphate Binders Sales Data," 2022.
- World Health Organization, "CKD Epidemiology," 2021.
More… ↓
