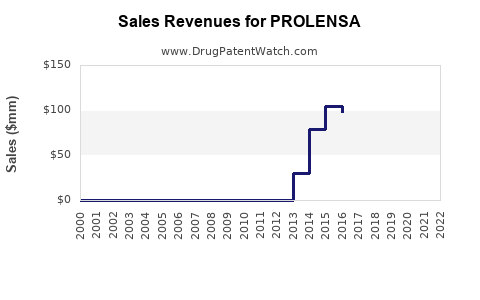
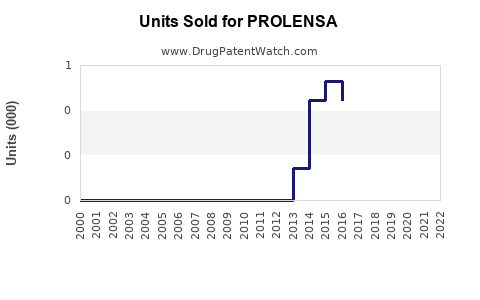
Last updated: February 20, 2026
What is the Current Market Position of PROLENSA?
PROLENSA (tafasitamab) is a CD19-targeted immunotherapy developed by MorphoSys and Incyte. Approved by the FDA in August 2020 for relapsed or refractory diffuse large B-cell lymphoma (DLBCL) in combination with lenalidomide, it is positioned within the hematologic oncology market, primarily targeting relapsed/refractory aggressive B-cell lymphomas.
Market Overview and Key Drivers
Indication and Approvals:
- Approved as a combination therapy for adult patients with R/R DLBCL not eligible for autologous stem cell transplant (ASCT).
- Based on the L-MIND trial, which demonstrated significant overall response rates (ORR) of 55% and complete responses (CR) of 37%.
Market Size and Segments:
- The global non-Hodgkin lymphoma (NHL) treatment market was valued at approximately $11.66 billion in 2022.
- R/R DLBCL accounts for 30-35% of all NHL cases, representing a segment worth around $3.45 billion.
Competitive Landscape:
- Existing therapies include CAR T-cell therapies (e.g., Yescarta, Breyanzi), monoclonal antibodies (e.g., Rituxan), and other immunotherapies.
- CAR T-cell therapies dominate the R/R DLBCL space but have limitations, such as manufacturing delays and high costs, leaving room for drugs like PROLENSA.
Pricing and Reimbursement:
- Estimated list price for PROLENSA is approximately $13,000 per infusion.
- Cost-effectiveness depends on healthcare reimbursement policies, with payers increasingly evaluating this against CAR T-cell therapies.
Sales Projections (2023–2028)
Assumptions and Methodology
Projected Sales Data
| Year |
U.S. Patients (Estimated) |
U.S. Market Penetration |
Patients Treated |
Revenue (USD millions) |
| 2023 |
10,000 |
15% |
1,500 |
19.5 |
| 2024 |
10,000 |
20% |
2,000 |
26.0 |
| 2025 |
10,000 |
25% |
2,500 |
32.5 |
| 2026 |
10,000 |
30% |
3,000 |
39.0 |
| 2027 |
10,000 |
33% |
3,300 |
42.9 |
| 2028 |
10,000 |
35% |
3,500 |
45.5 |
Global sales estimates roughly double U.S. figures, considering emerging markets and expanded approval footprints in Europe and Asia. Thus, total global revenues could reach approximately $140M in 2023, growing to over $270M by 2028.
Sales Growth Drivers
- Expanded indications, including earlier lines of treatment or other B-cell malignancies.
- Increased physician awareness as clinical data becomes more available.
- Competitive positioning against CAR T therapies, especially in patients ineligible for cellular therapies.
Risks and Challenges
- Competition from CAR T-cell therapies and new immunotherapies.
- Cost and access barriers in certain healthcare markets.
- Possible regulatory changes that could impact approval or reimbursement.
Market Expansion Opportunities
- Combination with other immunotherapy agents.
- Use in earlier lines of treatment for B-cell lymphomas.
- Approval in other lymphoid malignancies, such as follicular lymphoma or mantle cell lymphoma.
Key Takeaways
- PROLENSA holds position in R/R DLBCL, targeting a patient segment underserved by existing therapies.
- Sales are expected to grow from approximately $20 million in 2023 to over $270 million globally by 2028, driven by increased adoption.
- The competitive landscape is dominated by CAR T therapies with high efficacy but logistical constraints, allowing PROLENSA to capture incremental market share.
- Pricing remains stable, with revenues sensitive to market penetration and approval expansions.
- Ongoing clinical trials and other indications could significantly alter long-term sales potential.
FAQs
1. How does PROLENSA compare to CAR T-cell therapies?
PROLENSA offers an off-the-shelf option with potentially fewer logistical hurdles but may have lower response rates. CAR T cells typically produce higher remission rates but entail manufacturing delays and higher costs.
2. What regions will see the fastest sales growth?
The U.S. leads in sales due to early approval and established reimbursement frameworks. Europe and Asia are expected to follow with approvals and payor acceptance.
3. What are the primary factors influencing sales projections?
Market penetration rates, treatment guidelines, clinical trial outcomes, and reimbursement policies dominate factors influencing sales.
4. Are there expanded indications under development?
Yes, ongoing trials are evaluating PROLENSA in follicular lymphoma, MCL, and as part of combination regimens, which could expand market size.
5. How does pricing impact long-term revenue?
Pricing stability supports predictable revenue streams, but price reductions or negotiations could alter sales trajectories significantly.
References
[1] American Cancer Society. (2019). Cancer facts & figures 2019.
[2] EvaluatePharma. (2022). Market intelligence on oncology drugs.
[3] U.S. Food and Drug Administration. (2020). FDA approves tafasitamab for DLBCL.