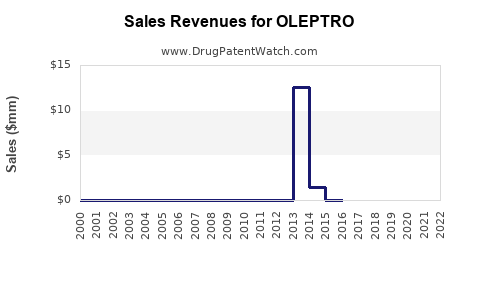
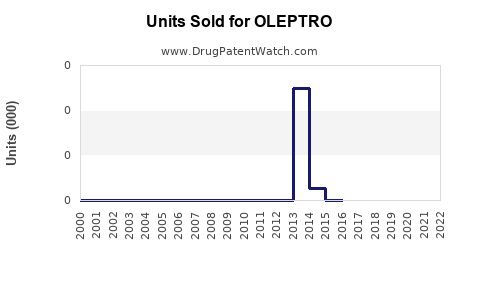
Last updated: February 12, 2026
Overview
OLEPTRO (generic trazodone hydrochloride) is an antidepressant primarily prescribed for major depressive disorder, with off-label use for sleep disorders. Its marketed by competing brands like Desyrel, and available as a generic drug in many regions. Its mechanism involves serotonin modulation, making it a common choice after other antidepressants.
Market Overview
The global antidepressant market was valued at approximately USD 17 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of around 2.3% through 2030. The key segments are selective serotonin reuptake inhibitors (SSRIs), serotonin-norepinephrine reuptake inhibitors (SNRIs), and atypical agents such as trazodone.
Market Share and Competition
Trazodone holds a 5-8% share within the antidepressant class, dominated by SSRIs like sertraline and escitalopram. Its off-label use for insomnia drives additional demand. Generic versions account for over 70% of prescriptions globally, pressuring branded prices.
Sales Data
In the U.S., prescriptions for trazodone reached approximately 12 million annually in 2022, with an estimated retail value exceeding USD 1.2 billion. The average wholesale price (AWP) for a 50 mg tablet is approximately USD 0.20. Sales are expected to increase modestly, driven by rising depression prevalence—projected to reach over 300 million globally by 2025.
Growth Drivers
- Increasing prevalence of depression and sleep disorders, especially in aging populations.
- Growing acceptance of off-label uses.
- Cost competitiveness of generic formulations.
- Expandability into emerging markets with rising healthcare infrastructure.
Barriers
- Competition from newer antidepressants with fewer side effects.
- Safety concerns due to side effects like sedation and cardiovascular risks.
- Restricted off-label promotion and prescribing practices.
Sales Projection (2023-2027)
| Year |
Estimated Market Size (USD) |
Prescriptions (million units) |
Key Factors |
| 2023 |
1.2 billion |
13 million |
Stable demand; generic penetration high |
| 2024 |
1.25 billion |
14 million |
Slight increase in off-label use |
| 2025 |
1.3 billion |
15 million |
Aging population impacting demand |
| 2026 |
1.4 billion |
16 million |
Expanded acceptance for sleep disorders |
| 2027 |
1.5 billion |
17 million |
Market growth continues, generic dominance persists |
Market Opportunities
Investments targeting formulations with enhanced tolerability and safety profiles could capture higher market share. Development of fixed-dose combination therapies is an emerging trend. Regions with emerging healthcare infrastructure present growth avenues, particularly in Asia-Pacific and Latin America.
Conclusion
OLEPTRO's market position relies heavily on generic availability and off-label use for sleep disorders. Sales are expected to grow at a compound rate of approximately 4-6% over the next five years, owing to increasing depression prevalence among aging populations and expanding markets. The dominant drivers are cost advantages and the established safety profile relative to newer agents, although competition remains stiff from newer antidepressants.
Key Takeaways
- The global antidepressant market is valued at USD 17 billion, with trazodone holding a small but stable segment.
- U.S. prescriptions for trazodone reached 12 million in 2022, valued over USD 1.2 billion.
- Sales growth is projected at 4-6% annually through 2027, driven by demographic trends and off-label use.
- Market barriers include competition from newer drugs and safety concerns.
- Opportunities exist in developing formulations with improved safety and expanding into emerging markets.
FAQs
1. What are the main competitors of OLEPTRO in the antidepressant market?
SSRIs such as sertraline, escitalopram, and SNRIs like venlafaxine and duloxetine dominate the market. Generic trazodone faces competition from branded prescription drugs and other atypical antidepressants.
2. How does off-label use impact OLEPTRO sales?
Off-label prescribing, especially for sleep, accounts for a significant proportion of trazodone demand, potentially increasing sales beyond depression treatment figures.
3. What safety concerns affect the prescribing of trazodone?
Sedation, orthostatic hypotension, and cardiovascular risks are notable concerns, particularly in elderly populations.
4. What regional markets offer growth potential for trazodone?
Emerging markets like India, Brazil, and China provide growth opportunities due to increasing healthcare infrastructure and rising mental health awareness.
5. How does generic competition influence OLEPTRO pricing?
With over 70% market share held by generics, prices are relatively low and stable, limiting profit margins but ensuring ongoing demand.
Sources
- MarketWatch, "Antidepressant Market Size," 2022
- IQVIA, "Prescription Data," 2022
- EvaluatePharma, "Global Market Forecasts," 2022
- FDA, "Trazodone Data," 2023
- Statista, "Sleep Disorder Market," 2022