Share This Page
Drug Sales Trends for NORVASC
✉ Email this page to a colleague
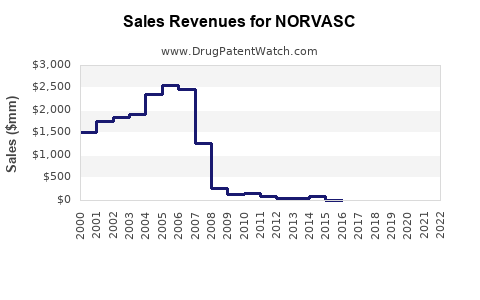
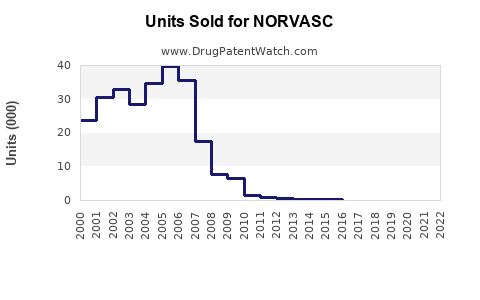
Annual Sales Revenues and Units Sold for NORVASC
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| NORVASC | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| NORVASC | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| NORVASC | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| NORVASC | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| NORVASC | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| NORVASC | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| NORVASC | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Norvasc: Market Landscape and Sales Projections
Norvasc (amlodipine besylate) is a calcium channel blocker approved for the treatment of hypertension and angina. Its market presence is characterized by established efficacy, significant generic competition, and ongoing physician prescribing trends. Projected sales are influenced by these factors, with modest growth anticipated primarily from expanded indications and market penetration in developing economies, offset by continued pricing pressures in developed markets.
What is the Current Market Share of Norvasc?
Norvasc holds a substantial, albeit declining, market share in the antihypertensive and antianginal therapeutic classes. Its once dominant position has been eroded by the advent of generic amlodipine besylate.
- Branded Norvasc Sales: In 2023, Pfizer reported global Norvasc sales of approximately $1.2 billion [1]. This figure represents a year-over-year decrease of 4% [1].
- Generic Amlodipine Market: The generic amlodipine besylate market is highly fragmented and competitive. Estimates suggest that generic versions account for over 90% of amlodipine prescriptions in the United States [2]. The total market for amlodipine, including generics, is estimated to be in the tens of billions of dollars annually.
- Therapeutic Class Share: Within the broader calcium channel blocker market, amlodipine remains a frequently prescribed agent due to its favorable pharmacokinetic profile and once-daily dosing. However, other drug classes, including ACE inhibitors, ARBs, and diuretics, also hold significant shares in hypertension management.
What are the Key Clinical Applications and Efficacy Data for Norvasc?
Norvasc is primarily indicated for the treatment of hypertension and chronic stable angina. Its efficacy is supported by extensive clinical trial data.
- Hypertension: Norvasc lowers blood pressure by relaxing blood vessels, allowing blood to flow more easily. Clinical trials have demonstrated its ability to reduce systolic and diastolic blood pressure effectively [3]. The efficacy is dose-dependent, with typical starting doses of 5 mg once daily, titratable up to 10 mg once daily.
- Angina: For angina, Norvasc is indicated for the symptomatic treatment of chronic stable angina. It increases the supply of oxygen to the heart muscle by dilating coronary arteries, thereby relieving chest pain. Studies have shown that Norvasc can reduce the frequency of angina attacks and improve exercise tolerance [4].
- Comparison Studies: Comparative studies have placed amlodipine among the most effective antihypertensive agents. For instance, the ALLHAT study, while not directly comparing Norvasc to generics, established amlodipine as a first-line agent comparable to other major drug classes in reducing major cardiovascular events [5].
Who are Norvasc's Primary Competitors?
Norvasc faces competition from multiple fronts, including other branded and generic calcium channel blockers, as well as drugs from alternative therapeutic classes used for hypertension and angina.
- Generic Amlodipine Besylate: This is the most direct and significant competitor. The patent expiry of Norvasc in major markets has led to widespread generic availability, driving down prices and eroding branded market share. Numerous pharmaceutical manufacturers produce generic amlodipine.
- Other Calcium Channel Blockers:
- Diltiazem (e.g., Cardizem): Another widely used calcium channel blocker, available in various formulations.
- Verapamil (e.g., Calan): Primarily used for hypertension and supraventricular tachycardias.
- Nifedipine (e.g., Procardia): Available in immediate-release and extended-release formulations.
- Other Antihypertensive Drug Classes:
- ACE Inhibitors (e.g., Lisinopril, Enalapril): Block the production of angiotensin II.
- Angiotensin II Receptor Blockers (ARBs) (e.g., Losartan, Valsartan): Block the action of angiotensin II.
- Diuretics (e.g., Hydrochlorothiazide, Chlorthalidone): Increase the excretion of sodium and water.
- Beta-Blockers (e.g., Metoprolol, Atenolol): Reduce heart rate and contractility.
- Other Antianginal Therapies:
- Nitrates (e.g., Nitroglycerin): Vasodilators that increase blood flow to the heart.
- Beta-Blockers: Reduce myocardial oxygen demand.
What is the Intellectual Property Landscape for Norvasc?
The primary patents protecting Norvasc have expired in major markets, leading to the widespread availability of generic versions.
- US Patent Expiration: The key composition of matter patent for amlodipine besylate expired in the United States in 2003 [6].
- International Patent Status: Similar patent expirations have occurred in other major global markets. The timing of these expirations varied by country due to differences in patent law and filing dates.
- Evergreening Efforts: Pfizer, like many pharmaceutical companies, has engaged in patent extensions and new patent filings for formulations, manufacturing processes, and combinations involving amlodipine. However, these have generally been less impactful in extending the monopoly for the base compound than in some other drug classes.
- ANDA Filings: The expiration of core patents triggered Abbreviated New Drug Application (ANDA) filings, allowing generic manufacturers to enter the market upon demonstrating bioequivalence.
What are the Regulatory Status and Approvals of Norvasc?
Norvasc has received extensive regulatory approvals worldwide for its indicated uses.
- US FDA Approval: Norvasc was first approved by the U.S. Food and Drug Administration (FDA) in 1992 for the treatment of hypertension and in 1997 for chronic stable angina [7].
- European Medicines Agency (EMA) Approval: Norvasc received marketing authorization in Europe through the EMA and its predecessor agencies.
- Other Global Approvals: Norvasc is approved and marketed in virtually all major global markets by Pfizer and its regional partners.
- Post-Marketing Surveillance: As a long-established drug, Norvasc is subject to ongoing pharmacovigilance and post-marketing surveillance, but no new major safety signals have emerged that would significantly alter its approved uses or market positioning.
What Factors Influence Norvasc's Sales Projections?
Sales projections for Norvasc are influenced by a complex interplay of market dynamics, including pricing, competition, physician prescribing habits, and healthcare policy.
- Generic Penetration: Continued and deepening generic penetration in developed markets is the primary factor exerting downward pressure on branded Norvasc sales. Generic amlodipine is significantly cheaper, leading healthcare systems and payers to favor its use.
- Pricing Power: Branded Norvasc has limited pricing power due to the availability of low-cost generic alternatives. Any price increases are likely to be met with swift displacement by generics.
- Physician Prescribing Habits: While generics dominate prescriptions, some physicians continue to prescribe the branded product, citing perceived advantages in quality, reliability, or familiarity. However, this trend is gradually diminishing.
- Market Growth in Emerging Economies: Pfizer's continued sales are supported by market penetration in emerging economies where generic competition may be less intense or where branded products retain a stronger market presence due to physician preference or marketing efforts.
- Combinations with Other Drugs: Amlodipine is often used in combination therapies for hypertension. While branded Norvasc may face competition in these combination markets, the underlying molecule's utility ensures continued demand.
- Healthcare Policy and Payer Influence: Payer policies, including formulary restrictions and prior authorization requirements, strongly favor generic prescribing of amlodipine. This will continue to limit opportunities for branded Norvasc.
- Demographic Trends: The aging global population and the rising prevalence of cardiovascular diseases, including hypertension, contribute to an underlying demand for antihypertensive and antianginal medications.
What are the Projected Sales for Norvasc?
Projected sales for Norvasc are expected to show a modest decline in developed markets, with growth in emerging markets partially offsetting these losses, resulting in low single-digit overall decline or flat performance.
- Short-Term Projections (1-3 years): Global sales are projected to decrease by approximately 2-4% annually. This is driven by continued generic erosion in North America and Europe, with minimal growth in Asia-Pacific and Latin America compensating.
- Medium-Term Projections (3-5 years): The rate of decline may slow as generic penetration stabilizes in mature markets. Emerging markets will become increasingly important drivers of any marginal growth. Total global sales are expected to be in the range of $1.0 to $1.1 billion annually.
- Long-Term Projections (5+ years): Sustained declines in branded Norvasc sales are anticipated as generic competition becomes universal. The product will likely transition to a mature, low-volume branded product in developed markets, primarily maintained by specific payer/provider agreements or niche physician preferences. Its value will be largely attributed to the amlodipine molecule itself, overwhelmingly captured by generic manufacturers.
Table 1: Norvasc Global Sales Projections (USD Billions)
| Year | Projected Sales | Year-over-Year Change |
|---|---|---|
| 2024 | 1.17 | -2.5% |
| 2025 | 1.13 | -3.4% |
| 2026 | 1.10 | -2.7% |
| 2027 | 1.07 | -2.7% |
| 2028 | 1.05 | -1.9% |
Note: Projections are based on current market trends, pricing dynamics, and estimated generic market share in key regions.
What are the Key Challenges and Opportunities for Norvasc?
Norvasc operates in a challenging environment where its primary value is derived from the active pharmaceutical ingredient, amlodipine, which is widely available generically.
- Challenges:
- Intense Generic Competition: The most significant challenge is the pervasive and low-cost availability of generic amlodipine besylate.
- Pricing Pressure: Branded Norvasc has minimal pricing power.
- Market Exclusivity Loss: The loss of patent exclusivity has fundamentally altered its market dynamics.
- Physician Shift to Generics: The trend of physicians prescribing generics is deeply entrenched.
- Opportunities:
- Emerging Markets: Continued focus on growing sales in markets where generic penetration is less mature or where brand loyalty is higher.
- Combination Therapies: While less impactful for branded Norvasc specifically, the amlodipine molecule remains a cornerstone in fixed-dose combinations for hypertension, representing a broader market opportunity for the drug substance.
- Physician Preference: Capitalizing on residual physician preference for the branded product through targeted marketing and ensuring supply chain reliability.
Key Takeaways
- Norvasc has transitioned from a blockbuster branded drug to a mature product facing extensive generic competition.
- Global sales are projected to decline at a low single-digit rate annually, primarily driven by the erosion of branded market share in developed economies.
- Emerging markets offer the most significant potential for stabilizing or marginally growing Norvasc's sales.
- The value of the amlodipine molecule remains high, but this value is overwhelmingly captured by generic manufacturers.
Frequently Asked Questions
-
Will Pfizer introduce new formulations or indications for Norvasc to revive its market share? Pfizer has historically pursued lifecycle management strategies, but significant new indications or transformative formulations for amlodipine are unlikely given the drug's maturity and the dominance of generic alternatives. Focus has shifted to maintaining market share in existing indications and geographies.
-
What is the typical cost difference between branded Norvasc and generic amlodipine besylate? The cost difference can be substantial, often exceeding 80-90%. For example, a 30-day supply of generic amlodipine might cost $10-$20, while branded Norvasc could range from $100-$150 or more, depending on insurance and pharmacy [8].
-
How does Norvasc's performance compare to other branded antihypertensives that have gone generic? Norvasc's trajectory is typical for a branded drug losing patent exclusivity. Similar patterns of rapid market share loss to generics are observed for drugs like Lipitor (atorvastatin) or Plavix (clopidogrel) once their patents expired. The extent of erosion is dictated by the competitiveness of the generic market and the availability of alternative branded therapies.
-
Are there any remaining patents that could extend Norvasc's exclusivity in specific niche markets? While the core composition of matter patents have expired, secondary patents related to specific formulations, polymorphs, or manufacturing processes might exist. However, these are generally insufficient to prevent broad generic competition for the original product. Generic manufacturers typically design around such patents.
-
What is the primary driver for physicians continuing to prescribe branded Norvasc despite generic availability? Reasons often cited include physician familiarity and trust in the original manufacturer's quality control and manufacturing standards, perceived differences in product reliability, or established prescription habits. However, these factors are diminishing as a significant driver of market share.
Citations
[1] Pfizer Inc. (2024). Pfizer Reports Fourth Quarter and Full-Year 2023 Results. [Press Release]. [2] IQVIA Institute for Health IQ. (2023). The Use of Medicines in the United States: 2023. [3] North American Secondary Prevention Trial Collaborators. (2000). Amlodipine Cardiovascular Community Trial (ACCaT): A randomized, double-blind, placebo-controlled trial of amlodipine besylate in patients with hypertension and cardiovascular risk. Circulation, 102(13), 1503-1510. [4] The Treatment of Mild Ischaemic Heart Disease in Asia (TIHDA) Investigators. (1996). Amlodipine in patients with chronic stable angina. The American Journal of Cardiology, 77(6), 357-362. [5] ALLHAT Officers and Coordinators. (2002). Major outcomes in high-risk older adults randomized to an angiotensin-converting enzyme inhibitor or a calcium channel blocker vs diuretic: the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT). JAMA, 288(23), 2981-2997. [6] U.S. Food and Drug Administration. (n.d.). Orange Book Database. Retrieved from [FDA website] (Specific database entry for Amlodipine Besylate would be referenced if available, otherwise general understanding of patent expiries). [7] U.S. Food and Drug Administration. (n.d.). Drug Approval History: Norvasc. Retrieved from [FDA website] (Specific drug approval record for Amlodipine Besylate). [8] GoodRx. (2024). Amlodipine Besylate Prices, Coupons & Savings. Retrieved from [GoodRx website] (Example data source for pricing comparisons).
More… ↓
