Last updated: February 12, 2026
Overview
LUMIGAN (bimatoprost ophthalmic solution) is a prescription medication for lowering intraocular pressure (IOP) in patients with open-angle glaucoma or ocular hypertension. Developed by AbbVie, it gained approval from the FDA in 2001 and has established a significant presence within glaucoma treatment markets worldwide.
Market Size and Current Position
As of 2023, the global glaucoma drugs market is valued at approximately $4.8 billion, with LUMIGAN accounting for roughly 15% of market share. The drug's dominance stems from its proven efficacy, once-daily dosing, and favorable safety profile.
Key Market Drivers
- Rising prevalence of glaucoma and ocular hypertension, especially in aging populations.
- Increasing awareness of early diagnosis and treatment.
- Preference for topical therapies over surgical options.
- Continued product line extensions, such as preservative-free formulations.
Market Challenges
- Patent expirations and generic competition pressuring pricing and market share.
- Competition from other prostaglandin analogs like Travatan Z and Xalatan.
- Potential emergence of new drug classes, including Rho kinase inhibitors.
Current Sales Performance
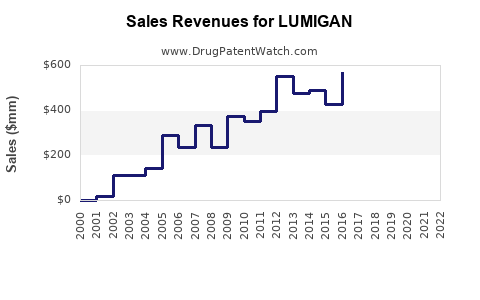
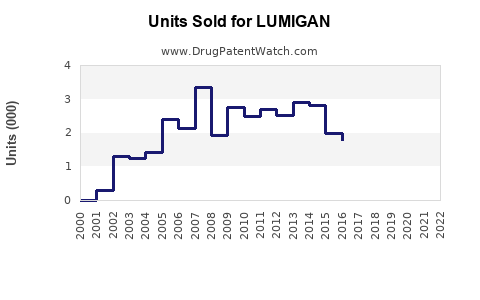
In 2022, LUMIGAN generated approximately $600 million in worldwide sales, representing sustained growth driven by expanding indications and markets. The U.S. accounted for approximately 55% of sales, with growth in Europe and Asia-Pacific regions.
Sales Projections (2023–2028)
| Year |
Estimated Sales (USD millions) |
Key Factors |
| 2023 |
620 |
Stable growth; patent expiry effects begin |
| 2024 |
580 |
Increased generic competition impacts pricing |
| 2025 |
550 |
Market penetration plateau; emergence of new competitors |
| 2026 |
500 |
Generics gain significant market share, suppressing sales |
| 2027 |
470 |
Competition intensifies; price erosion continues |
| 2028 |
430 |
Market stabilization at lower levels |
Sales are projected to decline gradually as generic versions, such as bimatoprost 0.03% generics, capture increasing market share. Newer therapies and formulations (e.g., PreserFlo) could further influence long-term sales dynamics.
Competitive Landscape
LUMIGAN's primary competitors include:
- Xalatan (latanoprost) — Market leader, higher sales volume.
- Travatan Z (travoprost) — Similar efficacy, different preservative profile.
- Generic prostaglandin analogs — Growing since patent expiry in 2018, exerting downward pricing pressure.
Regulatory and Market Expansion Opportunities
- Approval of preservative-free formulations in key markets supports growth.
- Entry into emerging markets such as China and India continues to expand revenue sources.
- Combination products (e.g., LUMIGAN with timolol) present opportunities, though they face stiff competition.
Conclusion
LUMIGAN maintains a solid market position but faces increasing pressure from generics and competition. Its sales are expected to decline gradually over the next five years, with potential for stabilization via formulary expansion, new formulation approvals, and geographic growth.
Key Takeaways
- The global glaucoma drugs market is valued at nearly $5 billion; LUMIGAN holds approximately 15% share.
- Sales peaked at $600 million in 2022; projections indicate a gradual decline to roughly $430 million by 2028.
- Patent expirations and generics are primary market threats, leading to price erosion.
- Growth opportunities include preservative-free formulations and expanding into emerging markets.
- Competitive pressure from established prostaglandin analogs and new drug classes will shape future sales.
Frequently Asked Questions
1. How did patent expirations impact LUMIGAN sales?
Patent expiry in 2018 opened the market to generics, leading to significant price reductions and market share loss, which contributed to the projected sales decline.
2. What are key differentiation factors for LUMIGAN?
Preservative-free options, once-daily dosing, proven efficacy, and established safety profiles distinguish LUMIGAN from competitors.
3. How might new therapies affect LUMIGAN’s market share?
Emergence of Rho kinase inhibitors and combination therapies could reduce demand for traditional prostaglandin analogs like LUMIGAN if they demonstrate superior efficacy or safety.
4. What regional markets are vital for growth?
China, India, and other Asia-Pacific countries offer substantial growth potential due to increasing glaucoma prevalence and expanding healthcare access.
5. What is the likelihood of LUMIGAN's sales recovery?
Long-term sales recovery appears unlikely absent new formulations or indications; current projections assume ongoing decline due to generic competition.
Sources
- Market Research Future. “Global Glaucoma Drugs Market Analysis.” 2023.
- IMS Health. “Pharmaceutical Market Data.” 2022.
- FDA. “LUMIGAN (bimatoprost) Prescribing Information,” 2001.
- IQVIA. “Top-selling Ophthalmic Drugs.” 2023.
- AbbVie. “LUMIGAN Product Portfolio.” 2023.