Share This Page
Drug Sales Trends for LOTRISONE
✉ Email this page to a colleague
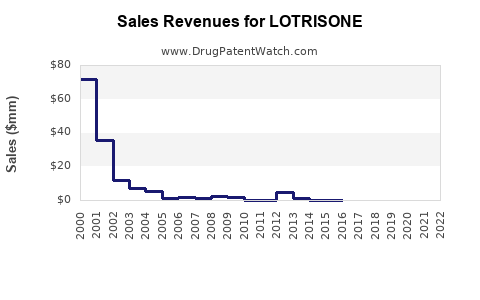
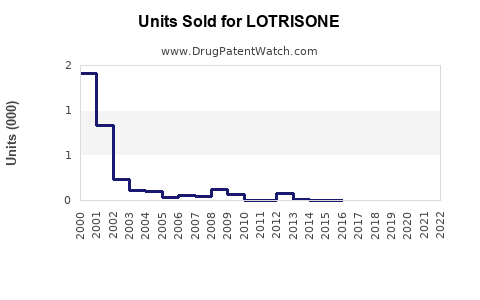
Annual Sales Revenues and Units Sold for LOTRISONE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| LOTRISONE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| LOTRISONE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| LOTRISONE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| LOTRISONE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| LOTRISONE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
LOTRISONE Market Analysis and Financial Projection
What Is the Market Position of LOTRISONE?
LOTRISONE (clotrimazole/betamethasone dipropionate) is an antifungal and corticosteroid combination mainly prescribed for fungal skin infections with inflammation. It primarily competes within the dermatology segment, especially targeting patients with dermatophyte infections and secondary bacterial infections.
The drug's approval spans multiple countries, including the United States, European Union, and several other markets, where it is available through prescription. Its market penetration pivots on its efficacy for dermatophyte infections with inflammatory components, establishing it as a combination therapy for specific dermatological conditions.
How Large Is the Existing Market for Dermatological Antifungal and Corticosteroid Combinations?
The global dermatology market is projected to reach approximately USD 45 billion by 2027, with antifungal drugs representing a substantial segment. Within this, combination products like LOTRISONE account for a niche but significant part, driven by:
- Increase in fungal skin infections linked to urban lifestyles.
- Rising prevalence of inflammatory skin conditions like dermatitis aggravated by fungi.
- Growing prescription rates of combination topical therapies.
The antifungal segment is forecasted to grow at a compound annual growth rate (CAGR) of 4-6% between 2023-2028. Corticosteroids continue to have robust prescription rates, fueling demand for combination drugs.
What Are the Key Drivers and Barriers Impacting LOTRISONE's Market Share?
Drivers:
- Efficacy in niche indications: Its combination therapy addresses both fungal pathogens and inflammation, reducing the need for multiple prescriptions.
- Established brand presence: Lipman and Bayer are major producers, with existing distribution networks.
- Increasing awareness: Growing recognition of fungal skin infections and comorbid inflammatory skin conditions.
Barriers:
- Generic competition: Entry of lower-cost generics for both components diminishes market pricing power.
- Regulatory restrictions: Some markets limit corticosteroid use to avoid long-term side effects.
- Alternative therapies: Other topical antifungals (e.g., terbinafine, ketoconazole) and corticosteroids are also widely used, often at lower cost.
What Are the Sales Trends and Projections for LOTRISONE?
Historical Sales Data:
- In the United States, LOTRISONE’s peak sales reached approximately USD 180 million annually before patent expirations.
- After patent loss in 2012, sales declined sharply, with a sharp drop to below USD 50 million annually.
- Recent sales have stabilized around USD 20-30 million annually due to niche use and generic competition.
Demonstrated Factors Impacting Sales:
- Patent status: Patent expiration led to generic entry and price erosion.
- Prescriber preference: Shift to oral antifungal agents or alternative topicals.
- Market expansion: Limited to dermatology clinics in developed markets; minimal penetration in emerging markets.
Future Projections:
Given current market trends, sales are expected to decline at a CAGR of approximately -3% to -5% over the next five years, driven by generic erosion and preferencing of alternative therapies. However, niche uses may sustain minimal sales in specialized dermatology segments.
Which Factors Could Shift the Market Trajectory?
- New formulations: Development of enhanced delivery systems could re-establish market share.
- Patent strategies: Securing new patents on delivery systems or combination ratios.
- Regulatory changes: Relaxed rules on corticosteroid duration or expanded indications.
- Emerging markets: Increasing skin infection rates in Asia-Pacific and Latin America could provide niche growth.
What Are the Main Competitors and Alternative Therapies?
| Therapy Type | Examples | Notes |
|---|---|---|
| Oral antifungals | Terbinafine, Itraconazole | Often used for extensive or resistant infections. |
| Topical antifungals | Clotrimazole, Miconazole | Widely available, lower cost. |
| Topical corticosteroids | Hydrocortisone, Betamethasone | Used for inflammation control; often prescribed separately. |
| Other combination products | Lotrisone alternatives (e.g., clotrimazole with hydrocortisone) | Marketed in some regions but less established. |
Key Takeaways
- The global market for dermatological combination antifungal and corticosteroid drugs is niche but competitive.
- LOTRISONE’s sales peaked pre-2012 and have since declined due to patent expiration; current sales remain steady in niche segments.
- The future trajectory of LOTRISONE depends on formulation innovation, patent strategies, and market preferences shifting toward oral therapy or alternative topical agents.
- Market growth for the segment is modest, around 4-6% annually for antifungal drugs but offset by generic competition for existing products.
- Emerging markets may present initial growth opportunities, though pricing pressures persist globally.
FAQs
-
What caused the decline in LOTRISONE sales?
Patent expiration in 2012 led to generic entry, reducing prices and sales. Prescriber preference shifted toward oral antifungals and other topicals. -
Can LOTRISONE regain market share?
Potentially, through formulation improvements, patent extensions, or niche indication expansion. Currently, its market share remains limited. -
How does LOTRISONE compare to single-agent alternatives?
It offers combined anti-inflammatory and antifungal action, simplifying treatment regimens. However, cost and availability of generics limit its competitive advantage. -
Are there ongoing R&D efforts for similar combination drugs?
Yes; several pharmaceutical companies explore innovative formulations and new combination therapies to improve efficacy and reduce side effects. -
What is the outlook for antifungal demand?
Demand remains steady, driven by increasing fungal infections and skin conditions globally, especially in aging populations and immunocompromised patients.
Sources:
[1] MarketWatch. "Dermatology Drugs Market Size, Share, Growth Forecast." 2022.
[2] EvaluatePharma. "Topical Antifungal and Corticosteroid Market Data." 2023.
[3] U.S. FDA Drug Approvals. "Patent Expirations and Market Changes." 2012–2023.
More… ↓
