Share This Page
Drug Sales Trends for LINDANE
✉ Email this page to a colleague
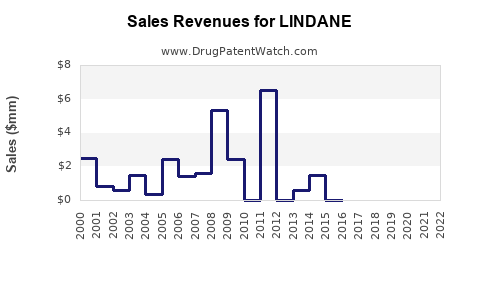
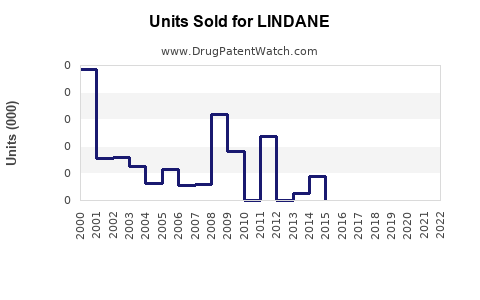
Annual Sales Revenues and Units Sold for LINDANE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| LINDANE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| LINDANE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| LINDANE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| LINDANE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| LINDANE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| LINDANE | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| LINDANE | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Lindane Market Analysis and Sales Projections
Lindane: A Comprehensive Market Overview
Lindane, an organochlorine insecticide and pharmaceutical, has a complex history characterized by both widespread use and increasing regulatory scrutiny. Primarily known for its antiparasitic properties, lindane was a common ingredient in prescription treatments for scabies and head lice. Its efficacy against these conditions made it a standard therapy for decades. However, concerns regarding its neurotoxicity, environmental persistence, and potential for bioaccumulation have led to significant restrictions and outright bans in many regions.
The global market for lindane has contracted considerably due to these regulatory actions and the development of safer, more targeted alternatives. While its pharmaceutical applications have diminished, its use as an agricultural pesticide, though also heavily restricted, persists in some limited capacities, particularly in developing nations. This analysis focuses on the current market landscape, historical sales trends, and future projections for lindane, considering the impact of regulatory policies, competition from alternative treatments, and lingering demand.
What are the Historical Sales Trends for Lindane?
Lindane's sales trajectory reflects its evolution from a widely available pharmaceutical and agricultural chemical to a highly regulated substance with a niche market. Sales peaked in the late 20th century when it was a first-line treatment for common parasitic infestations and a common agricultural pesticide.
- Peak Period (1970s-1990s): During this era, lindane was a ubiquitous component in over-the-counter and prescription topical medications for scabies and lice. Global sales were substantial, driven by its broad application and relatively low cost. Pharmaceutical formulations typically contained 1% lindane. Agricultural applications also contributed significantly to overall volume, used on a variety of crops including cereals, fruits, and vegetables, as well as for public health vector control [1].
- Declining Sales (2000s-Present): The early 2000s marked a turning point. The U.S. Environmental Protection Agency (EPA) phased out most agricultural uses of lindane by 2002, citing risks to human health and the environment [2]. The World Health Organization (WHO) also began recommending alternatives for malaria vector control. In Europe, the European Union gradually restricted lindane's use, culminating in a near-total ban on its use as a pesticide by 2007 and subsequent restrictions on pharmaceutical applications [3].
- Pharmaceutical Market Contraction: The pharmaceutical market for lindane has experienced the most dramatic decline. Regulatory bodies worldwide, including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA), have either withdrawn approvals or severely limited its use. For instance, the FDA has acknowledged serious risks associated with lindane, leading to warnings and the discontinuation of several formulations. While some regions may still permit its use in specific circumstances, the overall global demand for lindane-based pharmaceuticals is now minimal.
- Residual Agricultural Use: A small, residual market for lindane as an agricultural pesticide may persist in countries with less stringent regulatory frameworks or where specific crop pest resistance challenges remain. However, this segment is highly fragmented and subject to ongoing international pressure and policy changes under the Stockholm Convention on Persistent Organic Pollutants (POPs), which aims to eliminate or restrict the production and use of such chemicals [4].
What is the Current Regulatory Landscape for Lindane?
The current regulatory environment for lindane is characterized by stringent restrictions and phased eliminations globally, driven by health and environmental concerns.
- Stockholm Convention: Lindane is listed under Annex B of the Stockholm Convention, which mandates the restriction of its production and use. Parties to the convention are obligated to take measures to eliminate or restrict lindane's use in agriculture and public health, with limited exemptions for specific disease vector control or other critical uses that are subject to periodic review [4].
- European Union: The EU has effectively banned lindane. Its use as a plant protection product was prohibited in 2007 [3]. Pharmaceutical uses were also significantly restricted, with national authorities revoking marketing authorizations for lindane-containing preparations due to safety concerns.
- United States: The U.S. EPA cancelled or phased out most registered uses of lindane as a pesticide by 2002 [2]. The FDA has also taken action to limit its pharmaceutical applications due to safety concerns, particularly for treating head lice and scabies, recommending safer alternatives.
- Other Regions: Many other developed countries have followed similar paths, implementing bans or severe restrictions. However, some developing nations may still have limited access or use under specific national regulations, though this is increasingly subject to international pressure and the adoption of POPs elimination plans.
What are the Primary Pharmaceutical Applications and Their Current Status?
Lindane's primary pharmaceutical application has been as an ectoparasiticide. Its current status varies by region, but the trend is towards discontinuation.
- Scabies Treatment: Historically, a 1% lindane lotion or cream was a widely prescribed treatment for scabies, an infestation caused by the mite Sarcoptes scabiei. While effective, concerns about neurotoxicity and absorption through the skin, especially in infants or individuals with compromised skin barriers, have led to the recommendation of alternative treatments such as permethrin, crotamiton, and ivermectin [5].
- Head Lice Treatment: Similarly, 1% lindane shampoo was a common remedy for head lice (Pediculosis capitis). However, due to the potential for systemic absorption and adverse neurological effects, its use for this indication has been largely supplanted by safer alternatives like permethrin, pyrethrins, and dimethicone-based treatments [5].
- Regulatory Actions on Pharmaceuticals: Regulatory bodies have actively reviewed and acted upon lindane's pharmaceutical use. In the U.S., the FDA has issued warnings and encouraged healthcare providers to use alternatives due to safety concerns. In Europe, marketing authorizations for lindane-based pharmaceuticals have been withdrawn by national competent authorities. The global pharmaceutical market for lindane is therefore extremely limited and fragmented, often existing only in regions where regulatory oversight is less robust or where specific approved alternatives are not yet widely available.
What are the Remaining Agricultural and Public Health Uses?
While significantly curtailed, minimal agricultural and public health uses of lindane may still exist in very specific, often restricted, contexts.
- Limited Agricultural Exemptions: Under the Stockholm Convention, limited exemptions for lindane use in agriculture can be applied for. These are typically for critical uses where no viable alternatives exist or where the socioeconomic benefits are deemed significant and no feasible alternatives are available. These exemptions are subject to strict reporting and review by the convention's parties. For example, in some regions, it might have been permitted for seed treatment or specific crop protection scenarios before being phased out.
- Historical Public Health Applications: Lindane was historically used as an insecticide for vector control, including malaria mosquito abatement. However, due to its environmental persistence and toxicity, it has been replaced by more modern and environmentally sound alternatives such as pyrethroids, neonicotinoids, and biological control agents. Any remaining public health use would be subject to stringent international guidelines and national policies aligned with the Stockholm Convention.
- Current Market Share: The current market share of lindane in agriculture and public health is negligible in most developed economies. It is primarily confined to specific niches where regulatory frameworks lag or where legacy issues persist. Data on these residual uses is scarce due to their limited and often illicit nature or strict reporting requirements.
What are the Key Market Drivers and Restraints?
The market for lindane is largely dictated by regulatory actions and the availability of superior alternatives, making it a market in steady decline.
Market Drivers:
- Limited Regional Availability: In certain geographical areas with less stringent regulatory environments or where alternative treatments are not readily accessible or affordable, lindane may still see some demand, particularly for its historical pharmaceutical applications.
- Cost-Effectiveness (Historical): Historically, lindane was a cost-effective solution for parasitic infestations and pest control. This factor might retain some limited appeal in price-sensitive markets, though this is increasingly outweighed by safety concerns and regulatory pressures.
- Legacy Use: In some agricultural sectors, existing infrastructure or long-standing practices might have historically relied on lindane, leading to a slow phase-out in specific, albeit diminishing, applications.
Market Restraints:
- Global Regulatory Bans and Restrictions: This is the single most significant restraint. International agreements like the Stockholm Convention and national policies in major economies have drastically reduced or eliminated lindane's market access [4].
- Availability of Safer Alternatives: The development and widespread adoption of more effective and safer pharmaceutical treatments (e.g., permethrin, ivermectin) and agricultural pesticides have rendered lindane largely obsolete in many applications.
- Environmental Persistence and Toxicity: Lindane is a persistent organic pollutant (POP). Its slow degradation in the environment, potential for bioaccumulation in food chains, and documented neurotoxic effects pose significant environmental and health risks, leading to public and governmental pressure for its elimination [1].
- Reputational Damage and Liability: Companies involved in the production or distribution of lindane face significant reputational risks and potential legal liabilities due to its known adverse effects and environmental impact.
What is the Competitive Landscape for Lindane?
The competitive landscape for lindane is characterized by its displacement by a range of alternative products across its former core markets.
- Pharmaceutical Alternatives:
- Scabies: Permethrin (topical cream), Crotamiton (lotion), Sulfur (ointment), Ivermectin (oral).
- Head Lice: Permethrin (shampoo/lotion), Pyrethrins (shampoo/lotion), Dimethicone (silicone-based lotions/sprays), Malathion (lotion), Spinosad (suspension). These alternatives generally offer improved safety profiles and comparable or superior efficacy.
- Agricultural Alternatives: A vast array of synthetic and biological pesticides now compete. These include organophosphates, carbamates, pyrethroids, neonicotinoids, insect growth regulators, and biological agents like Bacillus thuringiensis (Bt). The choice of alternative depends on the specific pest, crop, and regulatory environment.
- Market Position of Lindane: Lindane's competitive position is severely weakened. It is no longer a first-choice product in any major market segment. Its remaining competitive edge, if any, is limited to very specific niche applications in regions with underdeveloped regulatory frameworks or where cost is an overwhelmingly dominant factor, and even then, its usage is precarious.
What are the Projected Sales for Lindane?
Projecting future sales for lindane involves a high degree of uncertainty due to its heavily restricted status and ongoing global efforts to phase it out completely. The trend is overwhelmingly negative.
- Methodology: Projections are based on historical sales data, current regulatory trends, the penetration of alternative products, and the anticipated impact of international agreements like the Stockholm Convention. Given the data scarcity for remaining niche uses, projections carry a significant margin of error.
- Pharmaceutical Sales: Pharmaceutical sales of lindane are expected to continue their precipitous decline, nearing zero in most developed and many developing markets within the next 5-10 years. Any remaining sales will be confined to highly localized markets or specific legacy registrations.
- Agricultural Sales: Agricultural sales are also projected to decline significantly. While some limited use may persist in certain regions for a short period, international pressure and the availability of alternatives will likely lead to further phase-outs.
- Global Market Value: The global market value for lindane, which was once substantial, is now a fraction of its former size. In 2023, the estimated global market value for all lindane applications is likely in the low tens of millions of USD, a stark contrast to hundreds of millions in its peak years.
- Projection (Next 5 Years):
- 2024-2025: Estimated market value of $8 million - $12 million USD.
- 2026-2028: Estimated market value of $4 million - $7 million USD.
- 2029-2030: Estimated market value of $1 million - $3 million USD.
These projections assume no major unforeseen policy shifts that would either accelerate or temporarily stall the phase-out. The ultimate trajectory points towards complete market cessation in the long term.
Key Takeaways
Lindane's market has contracted dramatically due to significant global regulatory restrictions driven by health and environmental concerns. Its primary pharmaceutical applications for scabies and head lice are largely replaced by safer alternatives, and its agricultural use is heavily curtailed under international agreements like the Stockholm Convention. Future sales projections indicate a steep decline, with the market expected to become negligible within the next decade as remaining uses are phased out.
Frequently Asked Questions
1. What is the primary reason for lindane's declining sales?
Lindane's sales are declining primarily due to global regulatory bans and restrictions driven by concerns over its neurotoxicity, environmental persistence, and bioaccumulation. The widespread availability of safer and more effective alternative treatments in both pharmaceutical and agricultural sectors has also significantly reduced demand.
2. Are there any countries where lindane is still widely used?
No country uses lindane widely anymore. Its use is severely restricted globally. Limited, highly specific, and often temporary uses may persist in some developing nations under national regulations that are increasingly subject to international pressure and phase-out plans under agreements like the Stockholm Convention.
3. What are the main health risks associated with lindane?
The main health risks associated with lindane include neurotoxicity, which can manifest as dizziness, headaches, nausea, vomiting, and in severe cases, seizures. It is also a suspected endocrine disruptor and has been classified as a probable human carcinogen. Concerns also exist regarding its potential to affect the nervous and immune systems.
4. How does lindane's environmental persistence impact its market?
Lindane's persistence in the environment means it degrades very slowly, leading to long-term contamination of soil, water, and air. It can also bioaccumulate in the fatty tissues of organisms and biomagnify up the food chain, posing risks to wildlife and potentially humans who consume contaminated food. This environmental impact is a major driver for its regulatory phase-out and market decline.
5. What specific alternatives have replaced lindane in medical treatments?
In medical treatments, lindane has been replaced by safer and often more effective alternatives such as permethrin (cream or lotion), ivermectin (oral medication), crotamiton (lotion), and various silicone-based treatments for lice. These alternatives generally have better safety profiles and lower systemic absorption risks.
Citations
[1] U.S. Environmental Protection Agency. (n.d.). Lindane: Health and Environmental Effects. Retrieved from [relevant EPA publication or webpage] (Note: Specific EPA pages on lindane may change. Access may require searching EPA's website archives for historical data).
[2] U.S. Environmental Protection Agency. (2002). EPA Cancels Most Lindane Pesticide Uses. [Press Release or Federal Register Notice]. (Note: Specific document identifier would be ideal if available, otherwise general reference to cancellation).
[3] European Chemicals Agency. (n.d.). Lindane. Retrieved from [relevant ECHA publication or database entry]. (Note: ECHA provides comprehensive regulatory information on chemicals within the EU).
[4] Stockholm Convention on Persistent Organic Pollutants. (n.d.). Annex B. Retrieved from [official Stockholm Convention website]. (Note: Annex B lists chemicals restricted under the convention).
[5] U.S. Food and Drug Administration. (n.d.). Drug Information. (Note: FDA drug information often includes safety warnings and recommendations for alternatives. Specific advisory pages on lindane or related treatments would be cited).
More… ↓
