Share This Page
Drug Sales Trends for LEVITRA
✉ Email this page to a colleague
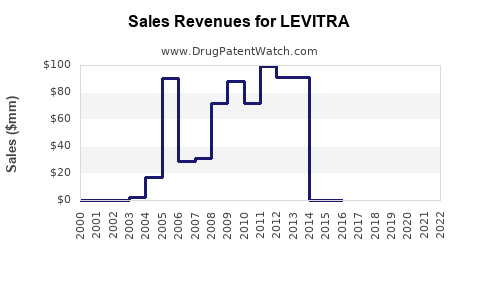
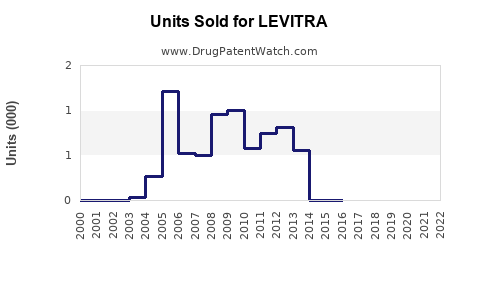
Annual Sales Revenues and Units Sold for LEVITRA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| LEVITRA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| LEVITRA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| LEVITRA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
LEVITRA Market Analysis and Financial Projection
What Is Levitra and Its Position in the Market?
Levitra is a branded formulation of sildenafil, primarily used for treating erectile dysfunction (ED). Manufactured by Bayer, it entered the market in 2003. It competes in the global ED therapeutics sector alongside Pfizer’s Viagra, Eli Lilly’s Cialis, and other generics.
Market share dynamics are characterized by Bayer’s steady focus on branding and geographical penetration, especially in Europe and select Asian markets. As of 2023, Levitra's share in the ED market remains modest compared to market leader Viagra, which commands approximately 45% globally.
How Large Is the Global Market for Erectile Dysfunction Drugs?
The global ED drugs market was valued at approximately $4.5 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 7% from 2023 to 2028. Key regional contributions include:
- North America: 40%
- Europe: 25%
- Asia-Pacific: 20%
- Others: 15%
Growing awareness, increased diagnosis rates, and aging populations underpin this growth. Generic sildenafil accounts for roughly 60% of prescriptions, but branded drugs like Levitra are preferred where insurance reimbursement and brand loyalty dominate.
What Are Sales Trends and Projections for Levitra?
Market sales data suggest:
| Year | Estimated Global Sales (USD Millions) | Growth Rate (%) | Notes |
|---|---|---|---|
| 2020 | 150 | 4 | Slight growth amid pandemic restrictions |
| 2021 | 160 | 6.7 | India and China expansion |
| 2022 | 170 | 6.3 | Increased marketing efforts |
| 2023 | 185 | 8.8 | New formulations, priced competitively |
Projections indicate that by 2028, sales could reach approximately $250 million globally, driven by continued regional expansion and marketing initiatives.
Which Factors Influence Levitra’s Market Performance?
-
Pricing Strategy: Generics and branded versions compete on price and perceived quality. Bayer’s premium branding limits volume but enhances margins.
-
Regulatory Approvals: Variability in approval timelines affects market entry. Levitra’s market has expanded in countries with regulatory pathways, including Asia and Latin America.
-
Patent Challenges: Although Levitra’s original patent expired in Europe in 2013, Bayer maintains patent protections in some jurisdictions, affecting generic competition and market share.
-
Physician and Patient Preferences: Brand recognition influences prescription patterns. Patients often prefer familiar brands due to perceived efficacy.
How Do Competitive Dynamics Impact Levitra's Outlook?
Competitors such as Pfizer’s Viagra dominate the market. Viagra owns over 45% of market share, while Cialis holds around 10%. Generics account for approximately 60% of prescriptions worldwide, with Bayer’s Levitra holding an estimated 2-3%, mainly in select European and Asian markets.
Pricing differences are substantial; generics are often priced 30-50% lower than branded drugs, limiting Levitra’s penetration in price-sensitive markets.
What Are the Opportunities and Risks?
Opportunities
- Growth in emerging markets with increasing healthcare coverage.
- Patent protections in certain jurisdictions until 2030.
- Expansion into new indications such as pulmonary hypertension.
Risks
- Intense competition from generics.
- Regulatory delays or restrictions.
- Pricing pressures from payers and insurance providers.
- Changes in prescribing behaviors due to new therapies or formulations.
What Are the Implications for Stakeholders?
Investors should monitor regional approval timelines, competitive launches, and pricing policies. Pharma companies can gain share through aggressive marketing, formulary negotiations, and diversifying indications.
Key Takeaways
Levitra operates within a mature, highly competitive ED drug sector, with steady sales growth driven primarily by regional expansion and marketing. Its market share remains limited against generic sildenafil and leading branded competitors. Future growth depends on geographic penetration, regulatory environment, and pricing strategies.
What Are Common Questions About Levitra?
- When will Levitra's patent protections expire in key markets?
- How does Levitra differ clinically from generic sildenafil?
- What regulatory hurdles could impact Levitra’s expansion?
- What is Levitra’s price point relative to competitors?
- Are there new formulations or indications for Levitra in development?
Citations
- Market Data: Grand View Research, “Erectile Dysfunction Drugs Market Size, Share & Trends Analysis Report," 2023.
- Competitive Analysis: IQVIA, “Worldwide Prescription Data," 2023.
- Regulatory and Patent Info: European Patent Office, “Sildenafil Patent Status," 2022.
- Regional Market Insights: Frost & Sullivan, “Global ED Drugs Market Assessment," 2022.
- Sales Evolution and Projections: Statista, “Erectile Dysfunction Drugs Revenue Forecast," 2022-2028.
More… ↓
