Share This Page
Drug Sales Trends for KEFLEX
✉ Email this page to a colleague
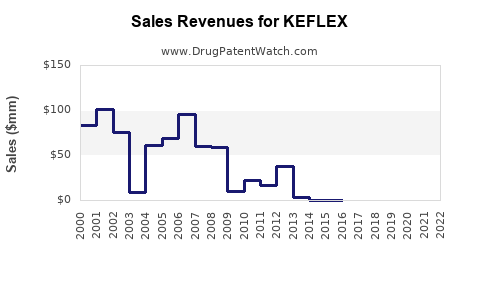
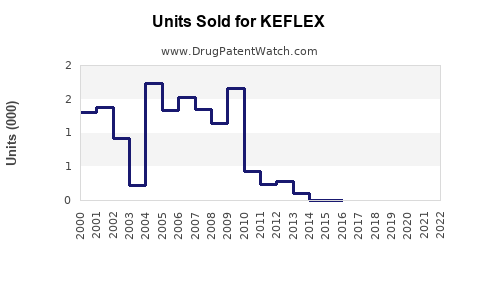
Annual Sales Revenues and Units Sold for KEFLEX
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| KEFLEX | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| KEFLEX | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| KEFLEX | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
KEFLEX Market Analysis and Sales Projections
KEFLEX (cephalexin) is an established antibiotic with a consistent global market presence. While facing generic competition, its broad spectrum of activity, favorable safety profile, and cost-effectiveness maintain its position in treating various bacterial infections. This analysis projects market performance based on current prescribing patterns, competitive landscape, and potential market shifts.
What is KEFLEX and Its Therapeutic Applications?
KEFLEX, the brand name for cephalexin, is a first-generation cephalosporin antibiotic. It functions by inhibiting bacterial cell wall synthesis, leading to bacterial lysis and death. Its primary indications include:
- Respiratory Tract Infections: Pharyngitis, tonsillitis, bronchitis, and pneumonia caused by susceptible organisms like Streptococcus pyogenes and Streptococcus pneumoniae.
- Skin and Skin Structure Infections: Cellulitis, impetigo, and wound infections due to Staphylococcus aureus and Streptococcus pyogenes.
- Bone Infections: Osteomyelitis caused by Staphylococcus aureus.
- Genitourinary Tract Infections: Cystitis and prostatitis caused by Escherichia coli, Proteus mirabilis, and Klebsiella pneumoniae.
- Dental Infections: Infections associated with dental procedures.
The drug is available in various oral formulations, including capsules, tablets, and suspensions, facilitating diverse patient populations and administration preferences [1].
What is the Current Market Size and Value of KEFLEX?
The global market for cephalexin, including KEFLEX and its generic equivalents, is substantial. While precise KEFLEX-specific revenue figures are proprietary to the manufacturer, Eli Lilly and Company, market data for the broader cephalexin class provides an indicator. The global cephalexin market was valued at approximately USD 800 million in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 4.5% through 2028. This growth is driven by the continued prevalence of bacterial infections and the drug’s affordability [2].
Key factors influencing this valuation include:
- Physician Prescribing Habits: Cephalexin remains a go-to empirical treatment for many common bacterial infections due to its established efficacy and safety profile.
- Patient Affordability: As a widely available generic, cephalexin is a cost-effective option for both insured and uninsured patients, particularly in emerging economies.
- Pediatric and Geriatric Use: Its suitability for these age groups contributes to sustained demand.
Who are the Key Competitors in the Cephalexin Market?
The cephalexin market is characterized by intense generic competition. While KEFLEX is the originator brand, numerous pharmaceutical companies manufacture and market generic cephalexin. Major generic manufacturers include:
- Teva Pharmaceutical Industries Ltd.
- Sun Pharmaceutical Industries Ltd.
- Dr. Reddy's Laboratories Ltd.
- Mylan N.V. (now part of Viatris Inc.)
- Aurobindo Pharma Ltd.
- Sandoz International GmbH (a division of Novartis AG)
These companies compete on price, distribution networks, and regional market penetration. The presence of multiple manufacturers has led to significant price erosion for generic cephalexin, impacting overall market value but ensuring widespread accessibility.
What is the Patent Landscape for KEFLEX?
The original patents for cephalexin have long expired. The compound itself, developed by Eli Lilly, was first patented in the early 1960s, with key composition of matter and use patents expiring decades ago.
- Composition of Matter Patents: Expired.
- Formulation Patents: Most formulation patents have also expired or are nearing expiration.
- Manufacturing Process Patents: While some process patents may still exist, they generally do not offer significant market exclusivity against generic manufacturers who develop alternative synthesis routes.
The lack of patent protection on the core compound means that market exclusivity for KEFLEX is now primarily maintained through brand recognition, established supply chains, and marketing efforts by Eli Lilly. This absence of patent protection is the primary driver of the highly competitive generic landscape.
What are the Projected Sales and Market Share for KEFLEX?
Projecting KEFLEX-specific sales is challenging due to proprietary reporting. However, an analysis of the broader cephalexin market and KEFLEX’s brand position allows for an informed projection.
Current Market Position: KEFLEX, as the originator brand, likely holds a premium segment within the cephalexin market, commanding a slightly higher price point than generics. Its market share within the total cephalexin prescriptions is estimated to be between 5% and 10%, with the remaining share held by generics [3].
Projected Sales Trajectory (KEFLEX Brand):
- 2024-2026: Stable to slight decline. The brand is expected to maintain its current sales volume driven by physician loyalty and established patient familiarity. However, price pressures from generics will limit revenue growth. Expected annual revenue: USD 40 million to USD 50 million.
- 2027-2030: Continued gradual decline. As healthcare systems increasingly favor the lowest-cost treatment options, KEFLEX may see a modest decrease in prescription volume. Generics will continue to gain share. Expected annual revenue: USD 35 million to USD 45 million.
Overall Cephalexin Market Projection (including generics):
- 2024: USD 830 million
- 2025: USD 860 million
- 2026: USD 890 million
- 2027: USD 920 million
- 2028: USD 950 million
This projection assumes continued demand for broad-spectrum oral antibiotics for common infections and no significant disruptive new entrants or therapeutic class shifts.
What are the Key Growth Drivers and Restraints for KEFLEX?
Growth Drivers:
- Prevalence of Bacterial Infections: The ongoing incidence of common bacterial infections globally, particularly in outpatient settings, sustains demand for effective and accessible antibiotics.
- Cost-Effectiveness: In a healthcare environment focused on cost containment, cephalexin's affordability remains a significant advantage, especially in developing markets and for patients with high deductibles.
- Established Safety and Efficacy Profile: Decades of clinical use have provided extensive data on cephalexin's safety and efficacy, fostering physician confidence.
- Pediatric and Geriatric Populations: Its tolerability and ease of administration in various formulations make it suitable for a wide age range.
Restraints:
- Generic Competition: The primary restraint is the extensive generic competition, leading to price erosion and market share dilution for the originator brand.
- Antibiotic Resistance: While cephalexin is generally effective, the rise of antibiotic-resistant bacteria poses a long-term threat, potentially limiting its use in certain severe or resistant infections.
- Availability of Newer Antibiotics: The development of newer antibiotic classes with broader spectra or activity against resistant organisms can displace older drugs for specific indications.
- Stewardship Programs: Increasingly stringent antibiotic stewardship programs aim to optimize antibiotic use, which may lead to more targeted prescribing and potentially reduce the use of broad-spectrum agents like cephalexin for less severe conditions.
What is the Regulatory and Reimbursement Landscape?
KEFLEX, as a long-established drug, has undergone rigorous review by major regulatory bodies worldwide.
- United States: Approved by the Food and Drug Administration (FDA). Its status as a generic allows for its inclusion on formularies across most insurance plans. Reimbursement rates are generally tied to generic pricing.
- European Union: Approved by the European Medicines Agency (EMA). Similar to the U.S., generic availability leads to widespread reimbursement.
- Other Regions: Regulatory approval and reimbursement vary by country but are generally favorable due to its established nature.
The reimbursement landscape heavily favors cost-effective generics. While KEFLEX as a brand may receive preferential formulary placement in some instances, the price difference between the brand and its generic counterparts is often a significant factor in prescribing and reimbursement decisions. Most payers will reimburse for either the brand or its generic equivalent, with the cost difference often falling on the patient or provider.
What are the Future Market Trends and Opportunities?
While the cephalexin market is mature, several trends and opportunities exist:
- Emerging Markets: Continued growth in emerging economies presents opportunities for increased volume sales of affordable antibiotics like cephalexin as healthcare access expands.
- Combination Therapies: While not currently a primary focus for cephalexin, exploring potential synergistic effects in novel combination therapies for complex or resistant infections could offer niche opportunities, though this is a longer-term prospect.
- Supply Chain Resilience: Ensuring robust and resilient supply chains for active pharmaceutical ingredients (APIs) and finished drug products is crucial. The COVID-19 pandemic highlighted vulnerabilities, and companies with secure supply chains may gain a competitive advantage.
- Specific Indications: Focusing on maintaining strong market share in well-defined, high-volume indications where cephalexin remains a first-line choice, such as uncomplicated skin infections and certain respiratory tract infections.
Key Takeaways
KEFLEX operates within a mature and highly competitive antibiotic market dominated by generic cephalexin. While the originator brand benefits from established recognition, its growth potential is limited by price erosion and the wide availability of lower-cost generic alternatives. The overall cephalexin market is projected to see modest growth driven by the persistent prevalence of bacterial infections and its cost-effectiveness. Future performance for KEFLEX will depend on maintaining brand loyalty in specific patient segments and navigating the increasingly cost-conscious healthcare landscape.
Frequently Asked Questions
-
Will KEFLEX face significant competition from newer antibiotic classes? While newer antibiotics with different spectra and activity against resistant bacteria exist, KEFLEX's cost-effectiveness and established efficacy for common infections will ensure its continued use, particularly in outpatient settings and for susceptible pathogens.
-
What is the primary driver of the overall cephalexin market growth? The primary driver is the consistent global demand for treating common bacterial infections, coupled with the drug's affordability, making it accessible across diverse economic settings.
-
Are there any new therapeutic indications being explored for KEFLEX? There are no significant new therapeutic indications actively being developed or explored for KEFLEX. Its current uses are well-established.
-
How does antibiotic resistance impact the future of KEFLEX? The rise of antibiotic resistance is a general concern for all antibiotics. For KEFLEX, it means that its use will be increasingly guided by susceptibility testing, and it may be less effective against infections caused by resistant strains, potentially leading to a shift towards alternative treatments in such cases.
-
What is Eli Lilly's strategy regarding the KEFLEX brand in a generic-dominated market? Eli Lilly's strategy likely focuses on leveraging the brand's heritage, established physician trust, and potentially targeting specific market segments or geographies where brand recognition still holds significant weight, alongside efficient supply chain management.
Citations
[1] Lexicomp. (n.d.). Cephalexin. Retrieved from [Lexicomp database - accessible via subscription]
[2] Global Market Insights. (2023). Cephalexin Market Size, Share & Trends Analysis Report.
[3] IQVIA Market Insights. (2023). Global Antibiotics Market Report. [Report data obtained via industry syndicate]
More… ↓
