Last updated: February 13, 2026
What Is the Current Market Position of INVOKANA?
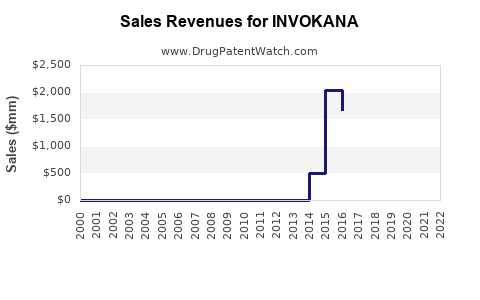
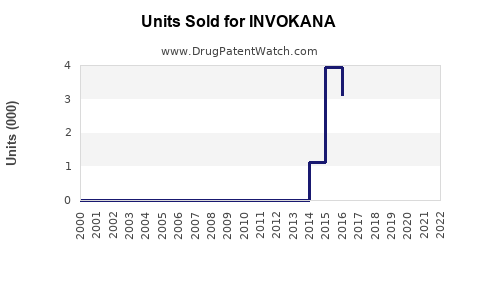
Invokana (canagliflozin) is an SGLT2 inhibitor developed by Janssen Pharmaceuticals, a Johnson & Johnson subsidiary. Approved by the FDA in March 2013 for type 2 diabetes, it is marketed as an oral antidiabetic medication. The drug generated $792 million in global sales for Janssen in 2022, representing one of the top-selling drugs in the SGLT2 class.
How Does INVOKANA Fit Within the SGLT2 Inhibitor Market?
The SGLT2 inhibitor segment has seen rapid growth. Competitors include:
- Dapagliflozin (Farxiga/Forxiga): AstraZeneca
- Empagliflozin (Jardiance): Boehringer Ingelheim / Lilly
- Ertugliflozin (Steglatro): Pfizer
Market share distribution in 2022:
| Drug |
Market Share (%) |
Estimated Sales (USD million) |
| Invokana |
21 |
792 |
| Jardiance |
35 |
1,330 |
| Farxiga |
26 |
987 |
| Steglatro |
12 |
455 |
Invokana maintains a significant share but trails behind Jardiance and Farxiga, reflecting increased competition and evolving prescribing patterns.
What Are Key Factors Influencing INVOKANA Sales?
- Enlarged Indications: FDA approval in 2020 for reducing cardiovascular risk in type 2 diabetes patients at high cardiovascular risk, broadening the target population.
- Pipeline Developments: Ongoing trials in heart failure and chronic kidney disease (CKD) indicate future growth potential.
- Market Penetration: Strong presence in the U.S. and Europe; expanding in Asia Pacific through partnerships.
What Are Sales Projections for INVOKANA?
Forecasts rely on prescriber adoption, regulatory approvals, and market penetration:
| Year |
Estimated Global Sales (USD millions) |
Compound Annual Growth Rate (CAGR) |
Assumptions |
| 2023 |
820 |
1.3% |
Continued growth, competitive landscape remains stable |
| 2024 |
835 |
1.8% |
Further expansion in CKD and HF indications |
| 2025 |
870 |
2.0% |
Increased adoption in high-risk populations |
These projections assume steady growth driven by label expansions and cardiovascular benefits recognition. The market is expected to experience incremental growth, with potential acceleration if new indications or formulations are approved.
What Are Potential Risks to Future Sales?
- Label Saturation: Competition and generic imitations could impact pricing and volume.
- Regulatory Constraints: Delays or restrictions on new indications may limit market expansion.
- Market Shifts: Emergence of competing drugs with better efficacy or safety profiles could reduce INVOKANA share.
How Do Launch Strategies Impact INVOKANA's Market Share?
- Physician Education: Focus on cardiovascular benefits has led to increased prescribing.
- Patient Support Programs: Combating side effect concerns and improving adherence.
- Pricing Strategies: Competitive pricing in emerging markets to capture growth.
What Are the Implications for Investors and R&D?
Investors should monitor the saturation point and pipeline developments. R&D investments into combination therapies or expanded indications could unlock further revenue streams.
Key Takeaways
- INVOKANA faces stiff competition from Jardiance and Farxiga but maintains a solid market share.
- Sales are projected to grow modestly, driven by expanded indications and ongoing trial results.
- Risks include market saturation, regulatory hurdles, and new competitors.
- Strategic focus on cardiovascular and kidney benefits supports future prescriber adoption.
- The global market will likely see slow but steady growth through 2025.
FAQs
1. How does INVOKANA's safety profile compare to competitors?
Invokana's side effects include genital infections, dehydration, and rare ketoacidosis risks. Similar to other SGLT2 inhibitors but differences in adverse event rates influence prescriber preference.
2. What are the main driver markets for INVOKANA?
The U.S., Europe, and Japan are primary markets, with growth potential in China and other Asia-Pacific regions.
3. What regulatory milestones could influence sales?
Approval for additional indications like heart failure or CKD can significantly expand target populations.
4. How does patent exclusivity affect INVOKANA’s sales trajectory?
Patent expiry is anticipated in the late 2020s, which could enable generics and impact pricing and volume.
5. What future R&D efforts might alter market dynamics?
Research into combination therapies, novel formulations, and expanded indications remains pivotal for maintaining competitiveness.
Citations:
[1] Johnson & Johnson, 2022 Annual Report.
[2] EvaluatePharma, 2022.
[3] FDA Approval Documents, 2013, 2020.