Last updated: February 12, 2026
What is Hydromorphone and its current market status?
Hydromorphone, marketed under brand names such as Dilaudid, is a potent opioid analgesic used for severe pain management. It is classified as a Schedule II controlled substance in the U.S., indicating high abuse potential. The drug functions primarily by binding to mu-opioid receptors in the central nervous system, providing analgesia.
The drug has a long-standing market presence, with generic formulations available globally. Its primary application is in hospitals, palliative care, and pain clinics. As of 2022, Hydromorphone's global market experienced steady growth driven by increasing chronic pain cases and surgical procedures, especially in North America and Europe.
What is the market size and current sales volume for Hydromorphone?
The global opioid analgesics market, including hydromorphone, was valued at approximately $8 billion in 2021 and projected to reach $11 billion by 2028, growing at a compound annual growth rate (CAGR) of 4.3%. Hydromorphone accounts for about 3-5% of this market, translating roughly to $240 million to $400 million in 2021.
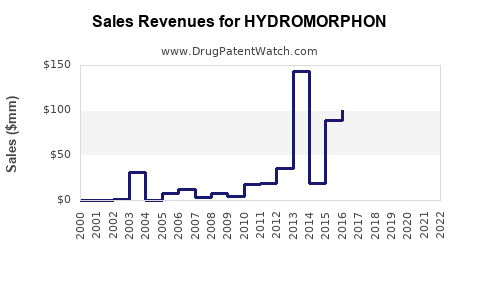
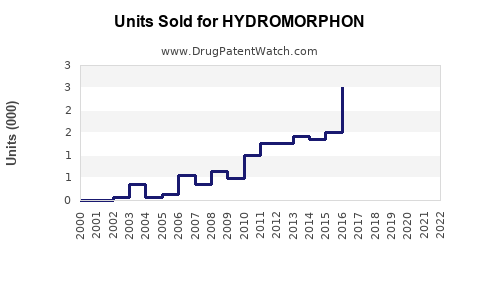
In the United States, Hydromorphone sales totaled $200 million in 2021, with hospital sales dominating demand. The drug’s prescription volume averaged around 2 million units annually, with a price per unit ranging from $50 to $100 depending on formulation and region.
How is the market expected to evolve over the next five years?
Market projections indicate a compound annual growth rate of approximately 3-5% for Hydromorphone, aligned with overall opioid analgesics market trends. Factors influencing growth include:
- Rising prevalence of chronic pain: The CDC estimates over 50 million adults suffer chronic pain, expanding demand.
- Surgical procedure volume: A projected 10% increase in surgeries annually elevates analgesic use.
- Regulatory environment: Stricter controls and opioid prescribing guidelines may constrict growth rates.
- Alternative therapies: The rise of non-opioid pain management options could suppress growth.
The U.S. is expected to maintain the largest share, driven by high opioid prescription rates, although scrutiny and disparities in prescribing practices could impact sales.
What are the key factors affecting future sales projections?
- Regulatory constraints: Increased oversight and potential reclassification can limit prescriptions.
- Opioid crisis impacts: Tighter regulations and shifts toward alternative treatments could reduce market size.
- Generic competition: The expiry of patents for brand-name formulations increases price competition.
- Healthcare trends: Growing emphasis on multimodal pain management may reduce dependence on opioids.
- International expansion: Emerging markets are beginning to adopt opioid-based analgesics, offering new revenue streams.
How competitive is the Hydromorphone market?
Mature in many regions, the market features several key pharmaceutical companies producing generic formulations. Major players include Pfizer, Teva Pharmaceuticals, and Sun Pharmaceutical Industries. Limited development efforts exist for new formulations, with most innovation focused on delivery methods, such as liquid or implantable forms.
Pricing strategies vary by region, with generics pushing prices downward. In the U.S., the average wholesale price (AWP) of Hydromorphone has decreased approximately 15% over the past five years amid increased generic penetration.
What are the regulatory trends and implications for sales?
In the U.S., the Drug Enforcement Administration (DEA) regularly reviews opioid classifications; recent proposals consider tighter controls. Some European countries have imposed stricter prescribing guidelines, impacting distribution.
Global regulatory trends forecast increased compliance costs and reporting requirements, potentially reducing supply chain efficiency. However, approved new formulations with abuse-deterrent features could sustain premium pricing and market share.
What sales strategies could optimize market penetration?
- Positioning hydromorphone as part of multimodal pain management protocols.
- Developing abuse-deterrent formulations to align with regulatory requirements.
- Targeting emerging markets with increasing healthcare infrastructure.
- Engaging in partnerships with hospitals for tailored dosing and delivery systems.
Key Takeaways
Hydromorphone’s market remains substantial, particularly within North America. Growth is steady but faces headwinds from regulatory pressures, the opioid epidemic, and competition from non-opioid therapies. Market expansion into emerging regions presents new opportunities, especially if formulations addressing misuse concerns are developed. Companies focusing on compliance, innovation, and expanded distribution channels will better position for sustained sales.
FAQs
1. How sensitive is Hydromorphone sales to regulatory changes?
Sales are highly sensitive. Stricter prescribing guidelines or reclassification can lead to demand reduction, while supportive regulations can facilitate market access.
2. Are there significant patent protections remaining for branded Hydromorphone?
Most formulations are off-patent, with current sales driven by generics, which suppress prices but ensure high volume.
3. What impact do non-opioid pain treatments have on Hydromorphone sales?
They pose a competitive threat; increasing adoption can reduce demand for opioid-based therapies.
4. How are counterfeit and abuse-deterrent formulations influencing the market?
Abuse-deterrent formulations can command higher prices and market share among prescribers concerned about misuse.
5. Is there potential for Hydromorphone in markets outside the U.S. and Europe?
Yes. Emerging markets are gradually adopting opioid analgesics, with differing regulatory and healthcare systems offering growth opportunities.
Sources:
[1] MarketWatch, "Opioids Market Analysis," 2022.
[2] IQVIA, "Global Pain Management Drugs Report," 2022.
[3] CDC, "Chronic Pain and Opioid Use Data," 2022.