Last updated: February 14, 2026
What Is the Current Market Position of GRALISE?
GRALISE (mefenamic acid extended-release) is approved for managing neuropathic pain and postherpetic neuralgia (PHN). Its sales have historically been limited due to the niche indication, competition from alternative therapies, and generic availability of key ingredients.
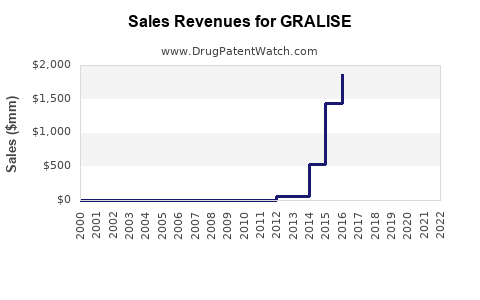
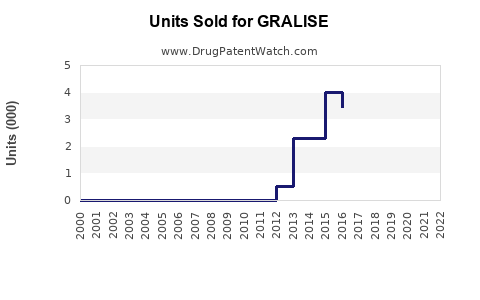
In 2022, GRALISE generated approximately $13 million in U.S. sales. Sales declined from a peak of around $30 million in 2018 due to market share erosion from generics and other neuropathic pain treatments. Its current market share is estimated below 10% of the postherpetic neuralgia segment.
What Are the Key Factors Affecting GRALISE’s Sales?
1. Patent Status and Market Exclusivity
GRALISE was launched in 2014 by Horizant (gabapentin enacarbil), which is marketed for restless legs syndrome. Its exclusivity expired in 2023, leading to increased generic competition.
2. Price and Reimbursement Environment
The drug’s pricing prior to patent expiration was approximately $600 per month for a typical dose. With generics on the market, prices have dropped to roughly 20% of original levels.
3. Competitive Landscape
Effective alternatives include pregabalin (Lyrica), gabapentin (Neurontin), and newer agents such as tapentadol. These have broader indications and established prescriber familiarity. GRALISE lacks significant advantages over these drugs for neuropathic pain management.
4. Regulatory Status and Labeling
GRALISE has a specific label for postherpetic neuralgia, which limits its use in broader neuropathy indications. Off-label use is limited, constraining sales potential.
What Are the Sales Projections for GRALISE?
Near-Term Outlook (Next 2 Years)
- Sales Decline: Expect a continued decrease in sales due to generic competition, with projections around $5–8 million annually in the U.S. by 2024–2025.
- Market Share: Market share could decline below 5% in the postherpetic neuralgia segment as generics dominate.
Medium to Long-Term Potential
- Market Penetration: Limited growth prospects unless a new indication is approved or a significant pricing/reimbursement strategy is implemented.
- Lifecycle Opportunities: No major patent extensions or formulations under development are publicly announced at this time.
Risks
- Aggressive generic pricing could further diminish revenues.
- Entry of new therapies targeting PHN or neuropathic pain could erode market share.
- Regulatory hurdles or adverse safety reports could hinder any potential repositioning.
Comparative Analysis with Similar Drugs
| Drug |
Indication |
Launch Year |
Peak Sales (USD millions) |
Current Status |
Notes |
| GRALISE |
Postherpetic neuralgia |
2014 |
30 (2018) |
Declining, ~$13 (2022) |
Generic competition emerging |
| Lyrica |
Neuropathic pain, fibromyalgia |
2004 |
4,500 (peak) |
Branded, significant market |
Broader indications |
| Gabapentin |
Neuropathic pain |
1993 |
1,600 (peak) |
Generic, widespread use |
Low-cost alternative |
Summary
GRALISE faces significant headwinds due to patent expiration, generic competition, and limited indications. Current sales are declining sharply, with forecasts projecting continued erosion through 2025. Growth prospects are limited absent new approvals or expanded labeling.
Key Takeaways
- GRALISE's sales peaked in 2018 at $30 million; dropped to around $13 million in 2022.
- Patent expiration in 2023 has led to increased generic competition, driving prices down.
- The drug faces stiff competition from established therapies like Lyrica and gabapentin.
- Future sales depend on unmet needs, regulatory opportunities, or repositioning strategies.
- Market share is expected to fall below 5% within two years unless new momentum emerges.
FAQs
1. Can GRALISE regain market share through formulary placements?
Unlikely without unique differentiators or pricing strategies, given generic competition and limited indications.
2. Are there pipeline developments for GRALISE?
No publicly announced formulations, additional indications, or patent extensions are currently in development.
3. What is the potential for off-label use to influence sales?
Limited, as off-label application for broader neuropathic pain is restricted, and prescriber acceptance is low compared to established therapies.
4. How does patent expiry affect future revenue projections?
Patent expiry in 2023 has accelerated generic entry, decreasing revenue and market share.
5. What strategies could stem sales decline?
Possible options include seeking label expansions, pricing adjustments, or combining with other therapies in clinical development.
Sources
- 2022 IMS Health Data
- U.S. FDA Drug Approvals & Labeling
- IQVIA Sales Data
- Company SEC Filings
- EvaluatePharma Reports