Share This Page
Drug Sales Trends for FEMHRT
✉ Email this page to a colleague
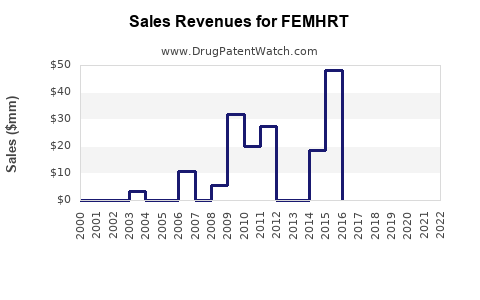
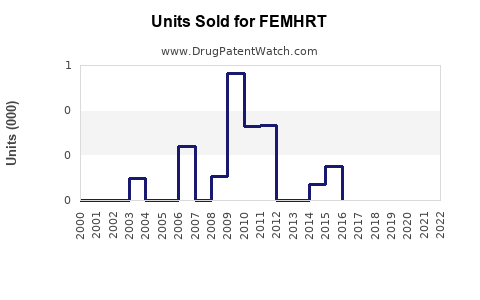
Annual Sales Revenues and Units Sold for FEMHRT
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| FEMHRT | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| FEMHRT | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| FEMHRT | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
FEMHRT Market Analysis and Financial Projection
What is FEMHRT?
FEMHRT is a hormone replacement therapy (HRT) drug containing a combination of estrogen (estradiol) and progestin (norethindrone) used primarily for managing menopausal symptoms and preventing osteoporosis in women with an intact uterus. Marketed by Pfizer, FEMHRT is available in oral tablet form.
What is the current market size for FEMHRT and comparable therapies?
The global hormone replacement therapy market was valued at approximately $1.8 billion in 2021 and is projected to reach $3.3 billion by 2028, expanding at a compound annual growth rate (CAGR) of roughly 8%.[1] The uptake of combination estrogen–progestin therapies constitutes about 70% of HRT prescriptions, driven by increased awareness of osteoporosis and menopause management.
North America holds over 45% of the global HRT market share, followed by Europe at approximately 30%. Demand is driven by aging populations, especially women aged over 50, and rising healthcare expenditure.
What are the key factors influencing FEMHRT sales?
- Regulatory approval cycles: Continued approval in major markets sustains sales. FEMHRT received FDA approval in 1994 and has maintained regulatory compliance.
- Prescribing trends: Physicians increasingly favor combination therapies with established safety profiles.
- Patient preferences: Oral formulations like FEMHRT are preferred for ease of administration.
- Competitive landscape: Canvas of generic options and alternative formulations, such as patches or gels.
How competitive is FEMHRT in its market segment?
Main competitors include:
| Product Name | Active Components | Dosage Forms | Market Status |
|---|---|---|---|
| Premarin | Conjugated estrogens | Vaginal, oral | Brand |
| EstroGel | Estradiol gel | Transdermal | Brand |
| Generic estrogen-progestin combos | Various | Oral | Generic |
FEMHRT's market share depends on brand loyalty and formulary inclusion. Its main advantage lies in its established safety data and clinical familiarity.
What are the sales projections for FEMHRT?
Projected sales depend on several factors:
- Market penetration: With about 20 million women in the US aged over 50, assuming 30% use HRT, the TAM (Total Addressable Market) is approximately 6 million women.
- Prescription rates: Current prescription rate estimated at 2 million annually.
- Market growth: With an 8% CAGR for HRT, prescriptions could reach 2.7 million annually within five years.
Assuming FEMHRT captures 10% of prescriptions initially, sales could approximate $150–200 million yearly, growing in line with market expansion and physician adoption. More conservative estimates peg FEMHRT’s share at 5–8%, translating to $75–120 million annually.
What are the pricing dynamics?
Average wholesale price (AWP) for FEMHRT is approximately $0.50 per tablet. With typical prescriptions of 1–2 tablets daily over 28 days, monthly revenue per patient is roughly $14–$28.
Considering discounts and rebates, actual net pricing may be 20–30% lower. This leads to annual revenue per patient between $170–$340. For a projected patient base of 200,000, annual sales could reach $34–$68 million initially.
What regulatory and patent considerations impact future sales?
- FEMHRT's primary patents expired in 2010, opening the market for generics.
- Patent litigation and exclusivity extensions may influence market entry timings for competitors.
- Regulatory updates regarding safety warnings for hormone therapies could affect prescribing patterns and sales.
What additional opportunities could influence FEMHRT sales?
- New formulations: Development of transdermal or topical versions could expand market reach.
- Combination therapies: Pairing with novel agents targeting osteoporosis or vasomotor symptoms.
- Market expansion: Entering emerging markets in Asia and Latin America offers growth potential.
Summary: Key Takeaways
- The global HRT market is projected to grow to over $3 billion by 2028, with estrogen-progestin therapies accounting for the majority.
- FEMHRT benefits from brand recognition but faces competition from generics and alternative delivery methods.
- Market penetration and physician prescribing behavior will determine sales volume.
- Conservative projections for FEMHRT sales range from $75 million to over $200 million annually within five years.
- Patent expiry and regulatory shifts could impact long-term market share.
FAQs
1. What are the primary drivers of HRT market growth?
Aging populations in developed markets, increased awareness of menopause management, and higher osteoporosis diagnosis rates.
2. How do regulatory changes affect FEMHRT sales?
Alterations in safety warnings or approval requirements can reduce prescription rates, while favorable labeling can stimulate demand.
3. Are there significant generic alternatives to FEMHRT?
Yes, numerous generic estrogen-progestin products are available, increasing price competition.
4. What role do patient preferences play in FEMHRT's market share?
Patients favor oral tablets for convenience, but transdermal options are gaining prevalence, impacting market dynamics.
5. How might emerging markets influence FEMHRT profits?
Growing healthcare infrastructure and aging populations in Asia and Latin America present new demand, provided regulatory pathways are addressed.
References:
[1] MarketsandMarkets, "Hormone Replacement Therapy Market," 2022.
More… ↓
