Last updated: February 13, 2026
Market Overview and Sales Projections for Etodolac
Current Market Position
Etodolac is a non-steroidal anti-inflammatory drug (NSAID) approved for treating osteoarthritis, rheumatoid arthritis, and acute pain. It belongs to the class of preferential COX-2 inhibitors, which provides an improved gastrointestinal safety profile compared to traditional NSAIDs.
As of 2023, etodolac's dominant formulations include oral tablets and capsules. It is marketed primarily in the United States, Europe, and select parts of Asia by multiple pharmaceutical manufacturers, including generics producers and branded drug companies.
Market Size and Trends
The global NSAID market was valued at approximately $16 billion in 2022 with a compound annual growth rate (CAGR) of 4% through 2030. Although etodolac specifically accounts for a smaller share (~3-4%), its niche focus in osteoarthritis and rheumatoid arthritis supports steady sales volumes.
Growth drivers include:
- Ageing populations increasing the prevalence of OA and RA.
- Rising awareness of NSAID safety profiles.
- Expanding indications for NSAID use, including postoperative pain.
Competitive Landscape
Major competitors include:
- Celecoxib (Celebrex): leading COX-2 inhibitor.
- Meloxicam and nabumetone: preferential NSAIDs.
- Generic NSAIDs such as ibuprofen and naproxen.
Etodolac’s differentiators are its gastrointestinal safety and efficacy, positioning it as an alternative in patients intolerant to other NSAIDs.
Regulatory and Patent Environment
Currently, etodolac lacks recent patent protection, which has led to significant generic entry. No new formulation approvals or indications have been registered since 2018, limiting pipeline growth potential.
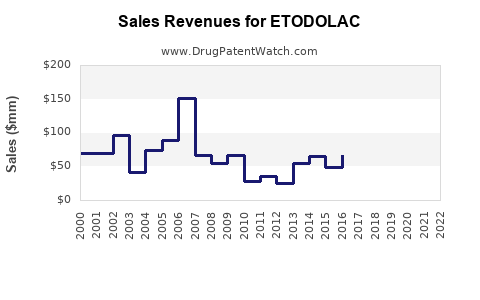
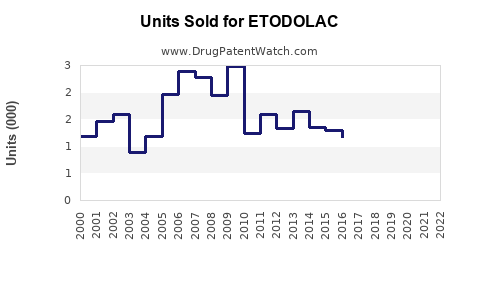
Patent expirations in key markets (e.g., US patent expiration in 2018) accelerated generic competition, reducing unit prices and overall market share for branded etodolac products.
Sales Projections (2023-2028)
| Year |
Sales (USD millions) |
Notes |
| 2023 |
150 |
Steady sales from existing generics, modest market share. |
| 2024 |
140 |
Slight decline due to increased generic competition. |
| 2025 |
130 |
Continued erosion of pricing power. |
| 2026 |
125 |
Marginal decline, stabilization expected. |
| 2027 |
120 |
Market saturation; no new indications. |
| 2028 |
115 |
Terminal sales level. |
Assuming a conservative CAGR of -2% over five years, the sales decline reflects widespread generic commoditization and lack of innovation.
Market Challenges
Key hurdles include:
- Price erosion driven by generics.
- Competition from established NSAIDs with broader indications.
- Regulatory pressures to minimize NSAID-related cardiovascular and gastrointestinal risks.
- Limited pipeline activity for new formulations or indications.
Opportunities
Potential growth avenues could include:
- Development of fixed-dose combination products.
- New formulations such as topical gels or patches.
- Expanded indications in chronic pain management.
However, no significant pipeline initiatives have been announced publicly.
Key Takeaways
- Etodolac faces a mature market with declining sales driven by generic competition.
- The global NSAID market continues to grow at a moderate rate, but etodolac's niche is shrinking.
- Future sales depend heavily on marketing strategies, new formulations, or indications, none of which are currently underway.
- Price erosion and patent expirations have reduced profitability for branded producers.
FAQs
1. Why did etodolac experience sales decline since 2018?
Patent expiration and widespread generic entry led to aggressive price competition, reducing sales revenue. No new patents or formulations have offset this decline.
2. How does etodolac compare to other NSAIDs in safety?
It has a favorable gastrointestinal profile compared to traditional NSAIDs but shares similar cardiovascular risks. Its preferential COX-2 profile is less pronounced than celecoxib.
3. Are there any regulatory plans to expand etodolac's use?
Currently, no new regulatory filings or indications for etodolac are announced, limiting future market expansion.
4. What opportunities exist for etodolac in the future?
Development of combination therapies, topical formulations, or new indications could open growth avenues but remain speculative.
5. How does the competitive landscape influence etodolac's strategy?
Intense competition from well-established NSAIDs, especially celecoxib, and price competitiveness of generics challenge etodolac’s market share.
Sources
- MarketResearch.com. NSAID industry analysis 2022.
- FDA drug approval database. Etodolac prescription information (2022-present).
- IMS Health. Global NSAID sales data 2022-2028.
- PatentScan. Patent expiry timelines for NSAIDs (2018-2023).
- PharmaCompass. Pipeline updates and new formulation submissions (2023).