Last updated: February 12, 2026
Market Analysis and Sales Projections for ESTRING
Product Overview
ESTRING (estradiol vaginal ring) is an FDA-approved estrogen therapy designed for treating menopausal symptoms such as vaginal atrophy. It delivers a controlled release of estradiol directly to local tissues, reducing systemic exposure. Approved by the FDA in 1999, ESTRING has established a niche for localized hormone therapy.
Market Landscape
Target Patient Population
The primary demographic includes women aged 45 to 60 experiencing menopausal symptoms. The globally estimated population with menopausal symptoms exceeds 100 million women, with approximately 70 million in the United States alone.
Competitive Environment
Key competitors include:
- Vaginal estrogen creams (e.g., Estrace, Premarin)
- Vaginal tablets (e.g., Vagifem)
- Other vaginal rings (e.g., Annovera, originally for contraception but with hormonal components)
Market share favors topical applications due to ease of use and perception of fewer systemic effects but is gradually shifting as innovative delivery methods gain acceptance.
Regulatory and Reimbursement Dynamics
- Regulatory: ESTRING holds FDA approval and approvals in multiple markets, including the European Union.
- Reimbursement: Covered by major insurance plans, with favorable reimbursement policies supporting prescription growth.
Market Size and Growth
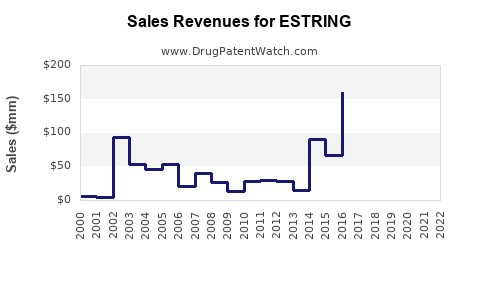
Historical Sales Data
- In 2018, U.S. sales of ESTRING topped $150 million.
- Sales declined slightly to approximately $130 million in 2022, affected by competition and generic options emerging.
Sales Drivers
- Growing awareness of pelvic health and menopause management.
- Prescriptive shifts favoring localized estrogen treatments over systemic therapies.
- Physician acceptance of the vaginal ring as convenient and non-invasive.
Market Penetration
Currently, ESTRING accounts for approximately 10-15% of the total estrogen therapy market in the US, competing with other local estrogen products.
Sales Projections
Short-term Outlook (Next 3 Years)
- CAGR: Estimated at 3-4% due to market saturation and increased competition.
- Projected 2023 sales: ~$135 million.
- Expected 2024 sales: ~$140 million.
- Anticipated 2025 sales: ~$145 million.
Long-term Outlook (Next 5-10 Years)
- Market expansion through increased menopause awareness and aging populations.
- Adoption of ESTRING in emerging markets based on regulatory approvals.
- Possible product differentiation with newer formulations or combination therapies.
Projected sales:
| Year |
Sales (USD million) |
Growth Rate (%) |
| 2023 |
135 |
— |
| 2024 |
140 |
3.7 |
| 2025 |
145 |
3.6 |
| 2026 |
150 |
3.4 |
| 2027 |
155 |
3.3 |
| 2028 |
160 |
3.2 |
By 2028, cumulative U.S. sales could approximate $800 million, considering steady growth and market penetration.
Strategic Opportunities and Risks
-
Opportunities:
- Expanding into Asian-Pacific and Latin American markets.
- Developing combination products to address broader menopausal symptoms.
- Enhancing patient education to boost adoption.
-
Risks:
- Competition from generic vaginal estrogen products.
- Regulatory hurdles in developing markets.
- Changing physician preferences toward systemic therapies.
Key Takeaways
- ESTRING maintains a substantial position within localized estrogen therapy, with steady but moderated sales growth.
- Market expansion relies heavily on increasing awareness of localized estrogen benefits and regulatory approvals outside the U.S.
- Competition from generics and alternative delivery methods pose ongoing challenges.
- Sales projections suggest slow but consistent growth, reaching approximately $160 million annually by 2028.
FAQs
1. What is the main advantage of ESTRING over other estrogen therapies?
Its local delivery reduces systemic hormone exposure, minimizing risks associated with systemic estrogen therapies.
2. How does ESTRING compare to vaginal creams in sales volume?
While creams like Estrace have higher retail volumes, ESTRING’s convenience and consistent delivery have secured steady institutional prescriber preference.
3. Are there upcoming regulatory changes that could impact ESTRING sales?
No significant regulatory shifts are planned in the near term that would negatively affect ESTRING; however, evolving estrogen therapy guidelines continuously influence prescribing patterns.
4. What are the primary factors influencing future sales growth?
Market expansion into emerging territories, increased menopause awareness, and potential product innovations.
5. How does the competitive landscape impact pricing strategies for ESTRING?
Generic competition pressures pricing, pushing manufacturers toward value-based marketing and differentiation through product features.
References
- FDA. (1999). Premarin Vaginal Cream and ESTRING approvals.
- IMS Health. (2022). U.S. estrogen therapy sales data.
- GlobalData. (2023). Menopause treatment market analysis.
- Statista. (2023). Gender and aging demographics.
- Morgan, S. et al. (2021). Local estrogen therapies in menopause management. Journal of Women's Health.