Share This Page
Drug Sales Trends for DOXYCYCLINE
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for DOXYCYCLINE (2020)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
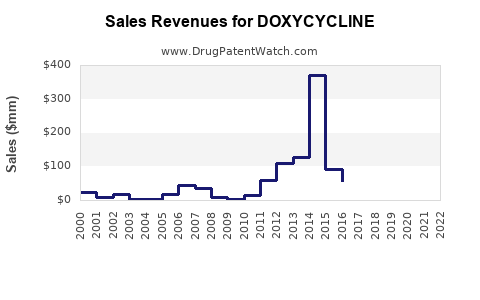
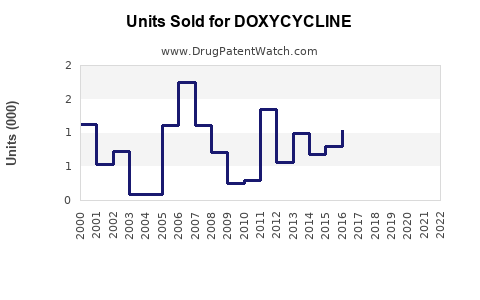
Annual Sales Revenues and Units Sold for DOXYCYCLINE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| DOXYCYCLINE | ⤷ Get Started Free | ⤷ Get Started Free | 2022 |
| DOXYCYCLINE | ⤷ Get Started Free | ⤷ Get Started Free | 2021 |
| DOXYCYCLINE | ⤷ Get Started Free | ⤷ Get Started Free | 2020 |
| DOXYCYCLINE | ⤷ Get Started Free | ⤷ Get Started Free | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Doxycycline Market Analysis and Sales Projections
Doxycycline, a tetracycline antibiotic, holds a substantial and growing market presence driven by its broad-spectrum efficacy, cost-effectiveness, and expanding therapeutic applications beyond infectious diseases. Current market valuation for doxycycline products, including generics and branded formulations, is estimated at $2.5 billion globally, with a projected compound annual growth rate (CAGR) of 4.5% over the next five years. This growth is underpinned by increasing incidence of bacterial infections, its established role in treating acne and rosacea, and emerging applications in areas such as malaria prophylaxis and anti-inflammatory therapies.
What is the Current Global Market Size for Doxycycline?
The global market for doxycycline generated approximately $2.5 billion in revenue in 2023. This figure encompasses various dosage forms, including oral tablets, capsules, and intravenous solutions, as well as both prescription and over-the-counter (OTC) products where legally permitted. The majority of this revenue is derived from generic formulations, which constitute over 80% of the market volume due to patent expirations of original tetracycline derivatives and the relatively low manufacturing costs associated with doxycycline production. Major contributing regions include North America and Europe, which account for roughly 60% of global sales, followed by Asia-Pacific, driven by a growing population and increasing access to healthcare.
What are the Key Drivers of Doxycycline Market Growth?
Several factors are contributing to the sustained growth of the doxycycline market:
- Prevalence of Bacterial Infections: Doxycycline remains a first-line or alternative treatment for a wide range of bacterial infections, including respiratory tract infections (e.g., pneumonia, bronchitis), urinary tract infections, skin infections (e.g., cellulitis), and sexually transmitted infections (e.g., chlamydia). The persistent global burden of these infections ensures consistent demand.
- Dermatological Applications: Doxycycline is a cornerstone treatment for moderate to severe acne vulgaris and rosacea. The increasing prevalence of these chronic skin conditions, particularly among adolescents and adults, fuels significant demand. The long-term nature of dermatological treatment protocols further contributes to recurring sales.
- Emerging Therapeutic Uses: Research and clinical application of doxycycline are expanding into non-infectious disease areas. Its anti-inflammatory and matrix metalloproteinase (MMP) inhibitory properties are being leveraged in:
- Malaria Prophylaxis: Doxycycline is a widely recommended and cost-effective option for preventing malaria in travelers to endemic regions.
- Periodontal Disease: It is used to reduce inflammation and inhibit collagenase activity in moderate to severe periodontitis.
- Rheumatoid Arthritis and Osteoarthritis: Ongoing research explores its potential to mitigate inflammation and cartilage degradation in these conditions.
- Cardiovascular Health: Studies suggest potential benefits in reducing inflammation associated with atherosclerosis.
- Cost-Effectiveness and Accessibility: As a generic drug, doxycycline offers a highly favorable cost-benefit profile compared to newer, patented antibiotics and treatments. This makes it an accessible and preferred option in both developed and developing healthcare systems, particularly in price-sensitive markets.
- Antibiotic Stewardship: While concerns about antibiotic resistance exist for all antibiotics, doxycycline's established efficacy and safety profile, coupled with its inclusion on the World Health Organization's List of Essential Medicines, maintain its position as a vital therapeutic agent when used appropriately.
What is the Competitive Landscape for Doxycycline?
The doxycycline market is highly fragmented, characterized by a large number of generic manufacturers and a few branded product holders. Competition is primarily based on price, product quality, and supply chain reliability. Key players include:
- Teva Pharmaceutical Industries Ltd.
- Sandoz (Novartis AG)
- Viatris Inc.
- Aurobindo Pharma Ltd.
- Dr. Reddy's Laboratories Ltd.
- Cipla Ltd.
- Zydus Lifesciences Ltd.
- Sun Pharmaceutical Industries Ltd.
These companies often compete on the basis of manufacturing scale, distribution networks, and the ability to offer multiple doxycycline formulations (e.g., immediate-release, delayed-release, extended-release). The market also includes smaller regional manufacturers catering to specific geographic demands.
What are the Projected Sales Figures for Doxycycline?
Based on current market trends, historical growth rates, and the identified growth drivers, the global doxycycline market is projected to reach approximately $3.1 billion by 2028, representing a CAGR of 4.5% from 2023 to 2028.
Projected Global Doxycycline Market Value (2023-2028) (in billions USD)
| Year | Market Value (USD Billion) |
|---|---|
| 2023 | 2.5 |
| 2024 | 2.61 |
| 2025 | 2.73 |
| 2026 | 2.86 |
| 2027 | 2.99 |
| 2028 | 3.12 |
This projection is supported by:
- Continued demand for infectious disease treatment: The ongoing prevalence of bacterial infections, coupled with the need for cost-effective treatments, will sustain a baseline demand.
- Growth in dermatological applications: The increasing incidence of acne and rosacea, and the long-term treatment cycles required, will continue to drive growth in this segment.
- Expansion of niche applications: The growing use of doxycycline for malaria prophylaxis and its exploration in chronic inflammatory conditions will contribute incremental growth.
- Generic market dynamics: The mature nature of the generic market implies price competition will remain, but volume growth will offset this, particularly in emerging economies with expanding healthcare access.
What are the Key Challenges and Risks in the Doxycycline Market?
Despite the positive outlook, several challenges and risks could impact the doxycycline market:
- Antibiotic Resistance: The overuse and misuse of doxycycline, like other antibiotics, can contribute to the development of antibiotic resistance. Growing resistance could limit its efficacy and lead to its restricted use in certain indications. Regulatory bodies and healthcare providers are increasingly emphasizing antibiotic stewardship programs.
- Competition from Newer Therapies: In specific therapeutic areas, particularly dermatology, newer classes of drugs with potentially different mechanisms of action or improved side-effect profiles are emerging. While doxycycline remains cost-effective, these alternatives could capture market share.
- Regulatory Scrutiny: Like all pharmaceuticals, doxycycline is subject to stringent regulatory oversight regarding manufacturing quality, efficacy, and safety. Changes in regulatory requirements or adverse event reports could impact market access or product approvals.
- Supply Chain Disruptions: The global nature of pharmaceutical manufacturing means that disruptions in raw material sourcing, production, or logistics can impact product availability. Geopolitical events, natural disasters, or trade disputes can pose risks.
- Pricing Pressures: The highly genericized nature of the doxycycline market leads to intense price competition. Manufacturers may face ongoing pressure to reduce prices, impacting profit margins.
- Side Effect Profile: While generally well-tolerated, doxycycline has known side effects, including gastrointestinal disturbances, photosensitivity, and tooth discoloration in developing children. These can limit its use in certain patient populations or lead to treatment discontinuation.
What is the Outlook for Doxycycline Formulations?
The market is expected to see continued dominance of oral solid dosage forms (tablets and capsules) due to their ease of administration, cost-effectiveness, and established manufacturing processes. However, there will be ongoing demand for:
- Extended-Release (ER) Formulations: These formulations offer improved patient compliance and potentially reduced side effects by maintaining more stable drug levels, particularly for dermatological applications.
- Intravenous (IV) Formulations: These are critical for hospitalized patients requiring rapid systemic delivery or when oral administration is not feasible due to severe illness or gastrointestinal issues.
- Pediatric Formulations: While the use of tetracyclines in young children is generally avoided due to tooth discoloration concerns, specific formulations may exist or be developed for older children or specific indications under strict medical supervision.
The development and approval of new doxycycline-based combination therapies or novel delivery systems could also influence market dynamics, though the core of the market will likely remain driven by established generic products.
Key Takeaways
- The global doxycycline market is robust, valued at $2.5 billion in 2023, with projections indicating growth to $3.1 billion by 2028.
- Key growth drivers include the persistent need for treatments for bacterial infections, strong demand in dermatology (acne, rosacea), and expanding therapeutic applications (malaria prophylaxis, anti-inflammatory uses).
- The market is characterized by a highly competitive generic landscape with significant price pressures.
- Potential challenges include the rise of antibiotic resistance, competition from newer therapies, and supply chain vulnerabilities.
- Oral solid dosage forms will continue to dominate, with ongoing demand for extended-release and intravenous formulations.
Frequently Asked Questions
-
What are the primary indications for doxycycline in 2024? Doxycycline's primary indications include a broad spectrum of bacterial infections such as respiratory, urinary tract, and skin infections, as well as acne vulgaris and rosacea. It is also used for malaria prophylaxis and in managing periodontal disease.
-
Which geographic regions represent the largest markets for doxycycline? North America and Europe constitute the largest markets for doxycycline, accounting for approximately 60% of global sales. The Asia-Pacific region shows significant growth potential.
-
What is the projected CAGR for the doxycycline market between 2023 and 2028? The projected compound annual growth rate (CAGR) for the global doxycycline market between 2023 and 2028 is 4.5%.
-
How does the emergence of antibiotic resistance impact the market for doxycycline? Antibiotic resistance poses a significant risk by potentially limiting the efficacy and broadening regulatory scrutiny on doxycycline's use. It necessitates a focus on antibiotic stewardship to preserve its therapeutic value.
-
Are there any significant patent expiries expected for doxycycline that could impact the market? As doxycycline is a well-established generic drug, the primary patents for its original development have long expired. The market competition is primarily driven by generic manufacturers, and significant new patent expiries for the molecule itself are not anticipated to drastically alter the competitive landscape beyond its already genericized status.
Citations
[1] World Health Organization. (2023). World Health Statistics 2023. Retrieved from https://www.who.int/publications/i/item/9789240075544 [2] National Institutes of Health. (2023). Doxycycline. LiverTox: Clinical and Toxicological Database. Retrieved from https://www.ncbi.nlm.nih.gov/books/NBK548901/ [3] Global Antibiotic Resistance and Surveillance System (GLASS). (2023). GLASS Report 2023. World Health Organization. Retrieved from https://www.who.int/teams/global-antimicrobial-resistance-and-stewardship/glass [4] Market Research Reports on Doxycycline. (Various Publishers, 2023-2024). Data compiled from leading pharmaceutical market intelligence firms including Mordor Intelligence, Grand View Research, and Allied Market Research, focusing on global antibiotic market trends and specific drug segment analyses. [5] U.S. Food & Drug Administration. (2023). Approved Drug Products Database. Retrieved from https://www.accessdata.fda.gov/scripts/cder/daf/
More… ↓
