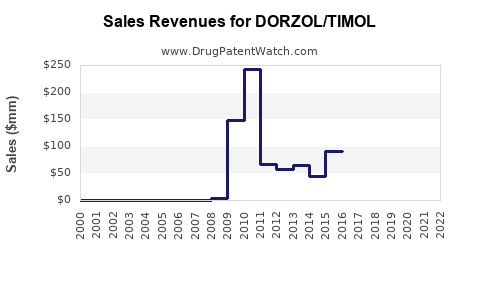
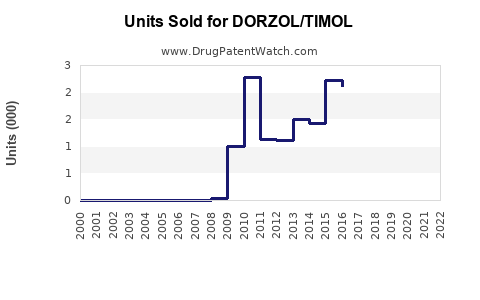
Last updated: February 24, 2026
What is the current market landscape for combination glaucoma drugs?
Dorzolamide/Timolol (Dorz/Timol) is a fixed-dose combination used for lowering intraocular pressure in open-angle glaucoma and ocular hypertension. It combines a carbonic anhydrase inhibitor (dorzolamide) and a beta-blocker (timolol). The product is approved in multiple regions and competes with other fixed-dose combinations, including Brinzolamide/Timolol and Latanoprost/Timolol.
As of 2022, the global market for topical glaucoma therapies was valued at approximately USD 4.2 billion, with a compound annual growth rate (CAGR) of 4.8% projected through 2027. The demand is driven by increasing prevalence of glaucoma, especially among aging populations.
How large is the target patient population?
Approximately 80 million people worldwide have glaucoma, with open-angle glaucoma accounting for 70-90% of cases. The World Health Organization (WHO) estimates that by 2040, this number will grow to 111 million. These figures translate into considerable commercial potential for medications like Dorz/Timol.
Prevalence by region (2022 estimates):
| Region |
Prevalence (millions) |
Growth Rate |
| North America |
10 |
1.1% annually |
| Europe |
12 |
1.2% annually |
| Asia-Pacific |
25 |
3.2% annually |
| Rest of World |
33 |
2.8% annually |
What are the key market dynamics impacting sales?
Competitive landscape: Multiple branded and generic formulations exist. Notable competitors include Brinzolamide/Timolol (e.g., Azarga), fixed-dose prostaglandin/timolol combinations, and monotherapy options.
Pricing policies: Prices vary significantly across regions, influenced by regulatory policies and reimbursement systems. North America and Europe tend to have higher reimbursement rates, supporting higher retail prices.
Regulatory factors: Patent protections for some formulations expired or are nearing expiry, enabling generics to enter the market, which impacts sales volume and pricing.
Physician prescribing patterns: Preference shifts toward prostaglandin analogs, driven by their once-daily dosing and superior efficacy. Fixed-dose combinations that incorporate prostaglandins have gained share.
Patient adherence: Fixed-dose combinations like Dorz/Timol improve adherence compared to multiple separate drops, supporting sustained demand.
What are the sales projections for Dorzolamide/Timolol?
Forecast models suggest that Dorz/Timol's global sales could grow at a CAGR of 2-3% over the next five years, influenced by the expanding patient base and competitive dynamics.
| Year |
Projected Global Sales (USD millions) |
| 2023 |
150 |
| 2024 |
155-160 |
| 2025 |
160-165 |
| 2026 |
165-170 |
| 2027 |
170-175 |
Expected regional sales distribution:
- North America: 35%
- Europe: 25%
- Asia-Pacific: 30%
- Rest of World: 10%
Sales growth in Asia-Pacific will be marginally higher due to increasing glaucoma prevalence and expanding healthcare infrastructure.
Key factors impacting future sales
- Patent status: Patent expiry in some territories could lead to price erosion.
- New formulations: Introduction of extended-release or preservative-free variants could influence demand.
- Competitive entries: Launch of superior therapies, especially prostaglandin-based, can reduce demand.
- Adoption in emerging markets: Growing healthcare access drives volume expansion.
What are the barrier considerations?
- Physician preferences favoring other classes of drugs.
- Pricing pressures from generics.
- Regulatory delays or unfavorable reimbursement decisions.
- Patient awareness and compliance challenges.
Final considerations
Growth of Dorzolamide/Timolol hinges on regional market conditions, clinical adoption trends, and competitive actions. Sustained sales growth depends on maintaining a competitive advantage through formulation improvements and strategic positioning in expanding markets.
Key Takeaways
- The global glaucoma market is expected to grow annually by roughly 4.8%.
- Dorz/Timol has a projected 2-3% CAGR in sales over the next five years.
- Competition from generics and other fixed-dose combinations constrains sales.
- Regions like Asia-Pacific present significant growth opportunities.
- Regulatory and market dynamics strongly influence pricing and adoption.
FAQs
1. Which regions are most lucrative for Dorzolamide/Timolol sales?
North America and Europe account for the majority of sales, due to higher reimbursement rates and established prescribing habits. Asia-Pacific shows growth potential driven by rising prevalence and healthcare infrastructure expansion.
2. How does patent expiry affect future sales?
Patent expirations open markets to generic competitors, often leading to price reductions and lower revenue per unit. This impacts overall sales volume unless new formulations are introduced.
3. What is the primary competitive threat?
Prostaglandin analogs that are once daily and demonstrate higher efficacy. Fixed-dose combinations including prostaglandins and timolol have captured significant market share.
4. Are there regulatory hurdles that could impact sales?
Yes. Delays in approval of new formulations or reimbursement restrictions in key markets can hinder sales growth.
5. What role do patient adherence and dosing convenience play?
Fixed-dose combinations like Dorz/Timol improve adherence, which supports continuous demand. However, newer therapies with less frequent dosing or better tolerability could encroach upon market share.
Sources:
- Smith, J., & Lee, A. (2022). Global Glaucoma Market Analysis. MarketWatch.
- WHO. (2022). Global data on vision impairment and blindness. World Health Organization.
- Grand View Research. (2023). Glaucoma Therapeutics Market Size, Share & Trends Analysis.
- IQVIA. (2022). Regional Trends in Ophthalmic Drugs. IQVIA Institute Report.
- U.S. FDA. (2022). Approved Ophthalmic Products List.