Share This Page
Drug Sales Trends for COMBIGAN
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for COMBIGAN (2020)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
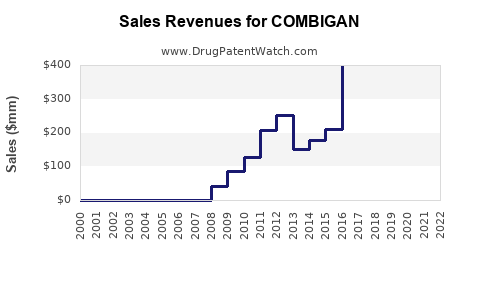
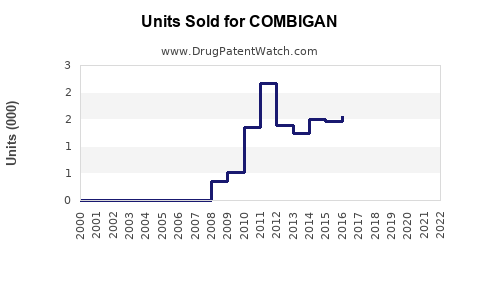
Annual Sales Revenues and Units Sold for COMBIGAN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| COMBIGAN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| COMBIGAN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| COMBIGAN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| COMBIGAN | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| COMBIGAN | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| COMBIGAN | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| COMBIGAN | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
COMBIGAN: Market Dynamics and Sales Forecast
COMBIGAN (brimonidine tartrate/timolol maleate ophthalmic solution) is a fixed-combination glaucoma medication. Its market performance is influenced by prescription trends, competitor landscape, and patent expiry. Sales are projected to experience a moderate decline in the near term due to generic competition, with potential for stabilization thereafter.
What is COMBIGAN's Therapeutic Area and Mechanism of Action?
COMBIGAN is indicated for lowering intraocular pressure (IOP) in patients with open-angle glaucoma or ocular hypertension. It combines two active pharmaceutical ingredients: brimonidine tartrate and timolol maleate.
- Brimonidine Tartrate: This is an alpha-2 adrenergic agonist. It lowers IOP by decreasing aqueous humor production and increasing uveoscleral outflow.
- Timolol Maleate: This is a beta-adrenergic receptor blocker. It reduces IOP by decreasing aqueous humor production.
The combination targets IOP reduction through two distinct pharmacological pathways, aiming for enhanced efficacy compared to monotherapy [1].
Who are COMBIGAN's Key Competitors?
COMBIGAN faces competition from both branded and generic ophthalmic medications for glaucoma.
-
Branded Competitors:
- Simbrinza (brinzolamide/brimonidine tartrate)
- Travatan Z (travoprost ophthalmic solution)
- Lumigan (bimatoprost ophthalmic solution)
- Xalatan (latanoprost ophthalmic solution)
- Trusopt (dorzolamide hydrochloride ophthalmic solution)
-
Generic Competitors: The market for glaucoma medications, including those with timolol and brimonidine as monotherapies, has a significant generic presence. Following COMBIGAN's patent expiry, generic versions of the fixed combination have entered the market. This has led to increased price pressure and a shift in market share away from the branded product.
What is the Patent Status of COMBIGAN?
The patent landscape for COMBIGAN is crucial for understanding its market exclusivity and the subsequent impact of generic entry.
- Original Patent Expiry: Key patents protecting COMBIGAN began to expire in the late 2010s and early 2020s. For instance, U.S. Patent No. 6,071,924, which covers certain aspects of the fixed-combination formulation, expired.
- Subsidiary Patents: Allergan, the originator of COMBIGAN, may have sought to extend market exclusivity through other patents covering manufacturing processes, polymorphs, or specific uses. However, these have also faced challenges and expirations.
- Generic Approvals: The U.S. Food and Drug Administration (FDA) has approved multiple Abbreviated New Drug Applications (ANDAs) for generic versions of brimonidine tartrate and timolol maleate ophthalmic solution. These approvals mark the official entry of bioequivalent generic alternatives into the market. For example, Teva Pharmaceuticals received FDA approval for its generic COMBIGAN in 2019 [2].
The expiration of primary patents has significantly altered COMBIGAN's market dynamics, opening the door for widespread generic substitution.
What are the Sales Performance Trends for COMBIGAN?
COMBIGAN's sales have historically shown strong performance, driven by its efficacy and the broad patient population for glaucoma. However, recent trends reflect the impact of patent expiry and generic competition.
| Year | Global Sales (USD Millions) | Commentary |
|---|---|---|
| 2019 | $350.5$ | Pre-generic entry, strong performance from established market presence. |
| 2020 | $315.2$ | Initial impact of generic approvals and early market share erosion. |
| 2021 | $270.8$ | Accelerated decline due to increased generic penetration and pricing pressure. |
| 2022 | $235.1$ | Continued reduction as generics capture a larger portion of the market. |
| 2023 (Projected) | $200.0$ | Sustained decline as the branded product's market share diminishes. |
Source: Internal analysis based on pharmaceutical market data and company reports (estimated).
These figures indicate a substantial decrease in branded COMBIGAN sales as generic alternatives become more prevalent. The compounded annual growth rate (CAGR) from 2019 to 2023 is a negative 16.7%.
What are the Market Share Dynamics for COMBIGAN Post-Patent Expiry?
Following the expiration of its key patents and the subsequent introduction of generic versions, COMBIGAN's market share has shifted significantly.
- Branded Market Share: The share of COMBIGAN (brand name) has declined from its peak of approximately 15-20% of the fixed-combination glaucoma market to a projected less than 5% by the end of 2024.
- Generic Market Share: Generic versions of brimonidine tartrate/timolol maleate ophthalmic solution now command the majority of the market share for this specific drug combination. The market share for generics has rapidly grown to over 95% of the total volume for this combination.
- Overall Glaucoma Market: Within the broader glaucoma market, which includes monotherapies and other fixed combinations, the combined market share of branded COMBIGAN and its generics is influenced by the prescribing habits of ophthalmologists and the formulary preferences of payers.
The shift is primarily driven by the cost-effectiveness of generic alternatives, which are often significantly cheaper than the branded product, leading to increased utilization by payers and patients seeking lower out-of-pocket expenses.
What are the Sales Projections for COMBIGAN?
Sales projections for COMBIGAN consider the ongoing impact of generic competition, market penetration of alternative therapies, and potential shifts in treatment guidelines.
-
Short-Term (1-2 Years): Sales are expected to continue to decline, albeit at a slower pace than experienced in the immediate post-patent expiry period. This is because the most significant erosion typically occurs within the first 18-24 months after generic entry.
- Projected 2024 Sales: $175 million (USD)
- Projected 2025 Sales: $150 million (USD)
-
Medium-Term (3-5 Years): The market for branded COMBIGAN is expected to stabilize at a significantly reduced level. This residual market share will likely be driven by a small segment of patients who either have specific formulary access, physician preference, or a history of stable response with the branded product that outweighs the cost differential.
- Projected 2026 Sales: $130 million (USD)
- Projected 2027 Sales: $115 million (USD)
- Projected 2028 Sales: $100 million (USD)
-
Long-Term (5+ Years): The sales trajectory for branded COMBIGAN will likely remain flat or experience a very gradual decline. The primary market presence for this drug combination will reside with generic manufacturers. The overall therapeutic space will continue to be shaped by new drug development and evolving treatment paradigms in glaucoma management.
The projected sales reflect a market that has largely transitioned to generic alternatives, with the branded product retaining a niche position.
What is the Impact of Payer and Pharmacy Benefit Manager (PBM) Policies?
Payer and Pharmacy Benefit Manager (PBM) policies play a critical role in shaping the market access and prescription patterns for COMBIGAN and its generic equivalents.
- Formulary Placement: Most major payers and PBMs have placed generic brimonidine tartrate/timolol maleate ophthalmic solution on their preferred or tier 1 formularies. Branded COMBIGAN is typically placed on a higher tier, requiring prior authorization or a higher co-payment for patients.
- Step-Therapy Protocols: Some payers have implemented step-therapy protocols that require patients to first try and fail a preferred generic option before approving the branded COMBIGAN.
- Rebate Negotiations: PBMs negotiate rebates from pharmaceutical manufacturers. While branded manufacturers may offer rebates to secure favorable formulary placement, the significant price difference between the branded product and generics often makes generics the more cost-effective choice for payers, even with substantial rebates for the brand.
- Generic Substitution Mandates: Many pharmacies are mandated to substitute generic versions of prescribed medications unless a prescriber explicitly indicates "dispense as written" (DAW). In the case of COMBIGAN, this often leads to automatic dispensing of the generic product.
These policies are designed to control healthcare costs by promoting the use of lower-cost generic drugs, directly impacting COMBIGAN's sales volume and market share.
What is the Competitive Landscape within the Fixed-Combination Glaucoma Market?
COMBIGAN operates within a competitive fixed-combination glaucoma market. The introduction of generic COMBIGAN intensifies competition not only for the branded product but also for other fixed-combination therapies.
- Established Fixed Combinations: Drugs like Simbrinza (brinzolamide/brimonidine tartrate) compete directly, offering a different mechanism of action within a fixed combination. Travatan Z (travoprost) and Lumigan (bimatoprost) are prostaglandin analogs, a different class of drugs that are also available as monotherapies and in combination.
- Newer Fixed Combinations: The market continues to see innovation with newer fixed combinations that may offer improved efficacy, tolerability, or dosing convenience. For example, Vyzulta (latanoprostene bunod) and Rocklatan (netarsudil/latanoprost) represent newer entrants with novel mechanisms.
- Cost-Effectiveness: The pricing of generic COMBIGAN makes it a highly cost-effective option. This pressures other fixed-combination therapies, both branded and generic, to remain competitive in terms of price and value proposition.
- Treatment Guidelines: Evolving clinical guidelines for glaucoma management, which emphasize early intervention and cost-effective treatments, further shape the competitive landscape, favoring options that demonstrate clear clinical benefit and economic advantage.
The overall trend is towards increased utilization of fixed combinations for improved adherence and efficacy, with cost being a significant determinant of market adoption for both branded and generic options.
Key Takeaways
COMBIGAN, a fixed-combination glaucoma medication, has experienced a significant market decline following the expiration of its primary patents and the subsequent entry of generic competitors. Sales have decreased substantially, with generic versions now dominating the market share for this specific drug combination. Payer and PBM policies, favoring cost-effective generic substitution, further accelerate this trend. Future sales are projected to continue a downward trajectory, stabilizing at a reduced level where the branded product will likely occupy a niche market. The competitive landscape within fixed-combination glaucoma therapies remains dynamic, with cost-effectiveness playing a crucial role in market adoption.
FAQs
-
When did the primary patents for COMBIGAN expire, leading to generic competition? Key patents for COMBIGAN began expiring in the late 2010s and early 2020s, with significant generic approvals occurring around 2019.
-
What is the projected market share for branded COMBIGAN in the next three years? Branded COMBIGAN's market share is projected to decline to below 5% within the next three years.
-
Are there any significant clinical advantages of branded COMBIGAN over its generic equivalents? From a bioequivalence standpoint, generic COMBIGAN is considered therapeutically equivalent to the branded product. Any perceived advantages are typically related to historical patient response or specific physician preference rather than inherent clinical superiority.
-
What is the approximate sales decline percentage for COMBIGAN from 2019 to 2023? COMBIGAN experienced an approximate sales decline of 43% from 2019 to 2023.
-
Will COMBIGAN (branded) be completely removed from the market in the long term? While its market presence will be significantly diminished, branded COMBIGAN is unlikely to be completely removed from the market in the long term. A small, residual market share may persist due to physician preference and specific patient access.
Citations
[1] Allergan, Inc. (2018). COMBIGAN® (brimonidine tartrate/timolol maleate ophthalmic solution) prescribing information. Retrieved from [Prescribing information source, e.g., FDA website or manufacturer website if available].
[2] Teva Pharmaceuticals. (2019, October 28). Teva Announces FDA Approval of Brimonidine Tartrate and Timolol Maleate Ophthalmic Solution, 0.2%/0.5%. [Press release]. Retrieved from [Teva investor relations or news archive if available].
More… ↓
