Share This Page
Drug Sales Trends for CLINDAMY/BEN
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for CLINDAMY/BEN (2020)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
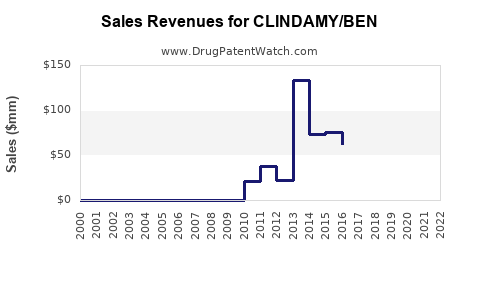
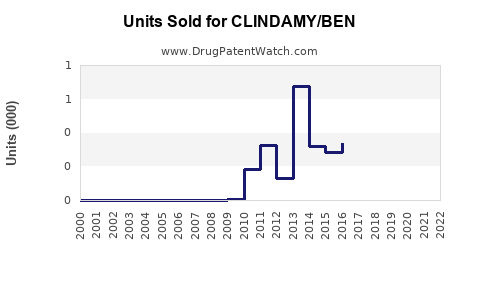
Annual Sales Revenues and Units Sold for CLINDAMY/BEN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| CLINDAMY/BEN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| CLINDAMY/BEN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| CLINDAMY/BEN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| CLINDAMY/BEN | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| CLINDAMY/BEN | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| CLINDAMY/BEN | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| CLINDAMY/BEN | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Clindamycin/Benzoyl Peroxide Topical Gel: Market Landscape and Sales Outlook
Clindamycin/benzoyl peroxide topical gel, primarily marketed under brand names such as Benzaclin, Duac, and BenzaClin, targets moderate to severe acne vulgaris. The combination therapy leverages clindamycin's antibiotic properties to reduce Cutibacterium acnes and benzoyl peroxide's antimicrobial and keratolytic effects. This analysis examines the current market, patent landscape, and projected sales for clindamycin/benzoyl peroxide topical gel.
What is the Current Market Size and Growth Trajectory for Clindamycin/Benzoyl Peroxide Topical Gel?
The global market for topical acne treatments, including clindamycin/benzoyl peroxide formulations, is substantial and projected to expand. This growth is driven by increasing acne prevalence, rising awareness of dermatological conditions, and a growing demand for effective cosmetic and therapeutic solutions.
- Global Acne Treatment Market: Valued at approximately $7.3 billion in 2022, the global acne treatment market is forecast to reach $13.8 billion by 2030, growing at a compound annual growth rate (CAGR) of 8.3% from 2023 to 2030 [1]. Topical treatments constitute a significant segment within this market.
- Clindamycin/Benzoyl Peroxide Share: While specific market share data for clindamycin/benzoyl peroxide topical gel alone is not consistently reported in broad market analyses, its established efficacy places it as a key player in the prescription topical antibiotic and antimicrobial segment for acne. Generic availability has increased accessibility, contributing to consistent market presence.
- Key Market Drivers:
- Prevalence of Acne: Acne vulgaris affects an estimated 80-90% of adolescents and a significant percentage of adults [2].
- Consumer Demand for Aesthetic Solutions: Increasing disposable incomes and a focus on personal appearance drive demand for effective acne treatments.
- Advancements in Formulations: Development of improved delivery systems and combination therapies enhances efficacy and patient compliance.
- Geographic Expansion: Growing healthcare infrastructure and awareness in emerging economies contribute to market expansion.
What is the Patent Landscape Surrounding Clindamycin/Benzoyl Peroxide Topical Formulations?
The patent landscape for clindamycin/benzoyl peroxide topical gel is characterized by foundational patents on the combination itself, followed by patents related to specific formulations, delivery systems, and manufacturing processes. The original composition patents have largely expired, leading to a market dominated by generic manufacturers. However, newer patents may cover novel aspects of the drug product.
- Original Composition Patents: The initial patents covering the combination of clindamycin and benzoyl peroxide in topical formulations have expired. For instance, patents related to early formulations of this combination have been in the public domain for years.
- Formulation and Delivery System Patents: Manufacturers may hold patents on specific ratios of active ingredients, excipients, pH adjustments, stabilizing agents, and unique delivery vehicles (e.g., specific gel matrices, encapsulation technologies). These can extend market exclusivity for branded products even after core composition patents expire.
- Example of Patent Focus: Patents may claim improved stability of clindamycin in the presence of benzoyl peroxide, enhanced penetration of active ingredients, or reduced skin irritation.
- Manufacturing Process Patents: Innovations in the scalable and cost-effective manufacturing of clindamycin/benzoyl peroxide gel can also be patentable, providing a competitive advantage.
- Expired Patents Impact: The expiration of key patents has facilitated the entry of generic versions. This has led to price competition and increased market penetration, but potentially lower revenue for individual manufacturers of branded products compared to periods of exclusivity.
- Search Strategy: A comprehensive patent search would typically involve databases such as the USPTO, Espacenet, WIPO's Patentscope, and Google Patents, using keywords including "clindamycin," "benzoyl peroxide," "topical," "gel," "acne," and relevant patent classification codes (e.g., A61K for preparations for medical, dental, or toilet purposes).
- Key Patent Holders (Historical and Current): Companies that have historically held or currently hold significant patents or market presence include but are not limited to Galderma (Duac), Valeant Pharmaceuticals (BenzaClin), and various generic manufacturers.
What are the Key Regulatory Approvals and Market Exclusivities?
Regulatory approvals from agencies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) are critical for market access. Patent status and regulatory exclusivities (like data exclusivity or market exclusivity) dictate the timeline for generic entry.
- FDA Approval: Clindamycin and benzoyl peroxide combinations have received FDA approval for prescription use in treating acne vulgaris. Specific product approvals (e.g., for Benzaclin, Duac) are listed in the FDA's Approved Drug Products With Therapeutic Equivalence Evaluations (the Orange Book).
- EMA Approval: Similar regulatory pathways exist in Europe through the EMA and national competent authorities.
- Generic Entry: Once patents expire and regulatory exclusivities lapse, generic manufacturers can seek approval to market bioequivalent versions of the drug. The FDA's Abbreviated New Drug Application (ANDA) process allows for this based on demonstrating pharmaceutical equivalence and bioequivalence.
- Market Exclusivity Types:
- Patent Exclusivity: Protection granted by patents.
- Data Exclusivity: A period where the FDA or EMA prohibits approval of generics based on the innovator's clinical trial data. This is distinct from patent protection.
- Market Exclusivity: Granted for specific circumstances (e.g., orphan drug exclusivity, pediatric exclusivity).
- Timeline Example (Hypothetical): If a foundational patent on a specific clindamycin/benzoyl peroxide formulation expired in 2018, and data exclusivity was for 5 years from the first FDA approval in 2013, a generic could have entered the market around 2018, assuming no other patent extensions or exclusivities were in place.
Who are the Major Market Players and Competitors?
The market for clindamycin/benzoyl peroxide topical gel includes both branded and generic pharmaceutical companies. Competition is driven by product efficacy, formulation differences, pricing, and market access.
- Branded Manufacturers:
- Galderma: Markets Duac® Gel.
- Bausch Health Companies Inc.: Markets BenzaClin® Gel.
- Generic Manufacturers: A significant number of generic pharmaceutical companies produce clindamycin/benzoyl peroxide topical gel. These include, but are not limited to:
- Teva Pharmaceuticals
- Apotex Inc.
- Sun Pharmaceutical Industries Ltd.
- Mylan N.V. (now part of Viatris)
- Actavis plc (now part of Teva)
- Competition Factors:
- Price: Generic competition significantly impacts pricing.
- Formulation: Minor differences in inactive ingredients or gel base can affect tolerability and patient preference.
- Distribution and Pharmacy Access: Wide availability through major pharmacies and healthcare providers is crucial.
- Marketing and Physician Detailing: Efforts to inform healthcare professionals about product benefits and new formulations.
What are the Sales Projections for Clindamycin/Benzoyl Peroxide Topical Gel?
Sales projections for clindamycin/benzoyl peroxide topical gel are influenced by the overall growth of the topical acne treatment market, the competitive landscape of branded versus generic products, and the introduction of new therapeutic agents. Given the established efficacy and generic availability, stable but moderate growth is anticipated.
- Projected Market Growth (for context): As noted, the broader acne treatment market is expected to grow at 8.3% CAGR.
- Clindamycin/Benzoyl Peroxide Specific Outlook:
- Unit Volume: Unit sales are expected to remain robust due to consistent demand for moderate-to-severe acne treatment and the cost-effectiveness of generics.
- Revenue: Revenue growth will be tempered by significant price erosion due to generic competition. Branded products may see flat to declining revenue unless they offer distinct formulation advantages. Generic revenue will depend on market share capture.
- CAGR Estimate (Clindamycin/Benzoyl Peroxide Segment): A CAGR of 3-5% for this specific combination product is a reasonable projection, balancing market expansion with generic price pressures. This would imply a global market value potentially reaching $1.5 billion to $2.0 billion within the next five years, assuming a conservative estimate of its share within the broader acne market.
- Factors Influencing Projections:
- New Entrants: Introduction of novel acne treatments (e.g., oral retinoids, targeted biologics, new topical agents) could shift treatment paradigms.
- Antibiotic Stewardship: Growing concerns about antibiotic resistance might lead to more judicious prescribing of topical antibiotics like clindamycin, potentially favoring benzoyl peroxide monotherapy or alternative non-antibiotic treatments for certain patient profiles.
- Insurance Reimbursement: Changes in formulary coverage and co-pay structures can impact patient access and prescription volume.
- Dermatologist Prescribing Habits: Continued preference for established, effective combinations like clindamycin/benzoyl peroxide.
What are the Key Challenges and Opportunities?
The market for clindamycin/benzoyl peroxide topical gel faces several challenges, but also presents opportunities for innovation and market penetration.
- Challenges:
- Antibiotic Resistance: Overuse or misuse of clindamycin can contribute to the development of resistant C. acnes strains, potentially reducing the long-term efficacy of the combination.
- Skin Irritation: Benzoyl peroxide, while effective, can cause dryness, redness, and peeling, leading to poor patient adherence.
- Generic Price Erosion: Intense competition from multiple generic manufacturers drives down prices and profit margins.
- Limited Differentiation: Branded products face challenges in differentiating themselves from generics unless significant formulation improvements are made.
- Opportunities:
- Improved Formulations: Developing gels with enhanced stability, reduced irritation, better patient compliance, and potentially improved penetration of active ingredients. This could involve novel excipients or delivery technologies.
- Combination Therapies: Exploring combinations with other anti-acne agents (e.g., retinoids, niacinamide) in fixed-dose formulations to offer broader spectrum efficacy and address multiple acne pathogenesis pathways.
- Targeted Delivery Systems: Research into microencapsulation or other advanced delivery methods to control the release of clindamycin and benzoyl peroxide, potentially reducing irritation and improving efficacy.
- Emerging Markets: Expanding access and physician education in developing regions where acne prevalence is high and effective, affordable treatments are needed.
- Patient Education: Implementing robust patient education programs to improve adherence and proper usage, thereby maximizing therapeutic benefit and mitigating resistance concerns.
Key Takeaways
The clindamycin/benzoyl peroxide topical gel market is a mature segment within the larger acne treatment landscape. While foundational patents have expired, leading to significant generic competition and price pressure, the product's established efficacy ensures sustained demand. Key growth drivers include the high prevalence of acne and increasing demand for dermatological solutions. Challenges revolve around antibiotic resistance and potential skin irritation, while opportunities lie in the development of novel formulations with improved tolerability and efficacy, and expansion into emerging markets. Projections indicate stable unit volume growth tempered by revenue moderation due to generic pricing.
Frequently Asked Questions
-
What specific acne severity levels is clindamycin/benzoyl peroxide topical gel indicated for? Clindamycin/benzoyl peroxide topical gel is primarily indicated for the topical treatment of inflammatory lesions of moderate to severe acne vulgaris [3].
-
What are the primary mechanisms of action for clindamycin and benzoyl peroxide in this combination? Clindamycin acts as an antibiotic, inhibiting the growth of Cutibacterium acnes bacteria implicated in acne pathogenesis. Benzoyl peroxide is an antimicrobial agent that kills C. acnes and also has keratolytic (exfoliating) and anti-inflammatory properties [4].
-
What are the common side effects associated with clindamycin/benzoyl peroxide topical gel? Common side effects include skin dryness, peeling, redness, itching, and burning sensation at the application site [3]. Severe side effects are rare but can include severe allergic reactions.
-
How does the development of antibiotic resistance impact the use of clindamycin/benzoyl peroxide topical gel? The emergence of clindamycin-resistant C. acnes strains is a concern. Prescribing guidelines often recommend combining clindamycin with benzoyl peroxide to help reduce the development of resistance, and limiting its use to inflammatory lesions [4].
-
Are there any significant differences between branded and generic clindamycin/benzoyl peroxide topical gels? While generic products must be bioequivalent to the branded product (demonstrating comparable absorption and distribution), there can be minor differences in inactive ingredients, formulation texture, and packaging. These differences typically do not affect the therapeutic efficacy or safety profile.
Citations
[1] Grand View Research. (2023). Acne Treatment Market Size, Share & Trends Analysis Report By Type (Topical, Oral), By Acne Type (Acne Vulgaris, Others), By Drug Class (Antibiotics, Retinoids, NSAIDs, Hormonal Therapy), By Distribution Channel, By Region, And Segment Forecasts, 2023 - 2030. Retrieved from https://www.grandviewresearch.com/industry-analysis/acne-treatment-market
[2] National Institutes of Health. (2022). Skin Conditions by the Numbers. Retrieved from https://www.niams.nih.gov/health-topics/skin-conditions (Note: While this is a general resource on skin conditions, specific prevalence data for acne is widely available from dermatological bodies and research publications.)
[3] U.S. Food and Drug Administration. (n.d.). Drug Database (Accessed for product-specific information on approved indications and side effects).
[4] Del Rosso, J. Q., & Alexis, A. (2017). Acne Vulgaris: A Review of Pathogenesis, Antibiotic Resistance, and Treatment Recommendations. Journal of Clinical Dermatology, 16(1), 11-19.
More… ↓
