Last updated: February 13, 2026
Market Overview
Carisoprodol is a muscle relaxant indicated for short-term management of acute musculoskeletal pain. Approved by the FDA in 1959, it has historically been prescribed in the United States and globally. Its core mechanism involves central nervous system depression, leading to muscle relaxation. Its-controlled substance status in multiple jurisdictions influences the market dynamic and regulatory landscape.
Regulatory Status and Impact
- In 2012, the FDA issued a boxed warning citing risks of dependence and withdrawal.
- The Drug Enforcement Administration (DEA) classified it as a Schedule IV controlled substance in 2012.
- Several countries, including the UK and Canada, have similar regulations restricting or monitoring its use, reducing prescribed volume.
- Prescription restrictions have led to decreased sales in some markets, while others maintain its status with limited regulatory adjustment.
Market Size and Historical Sales Trends
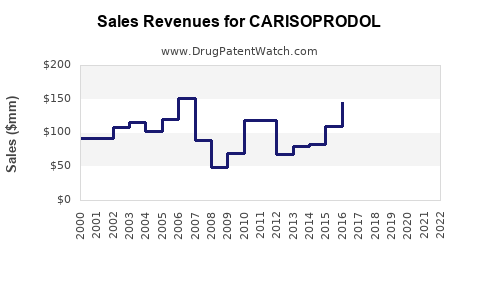
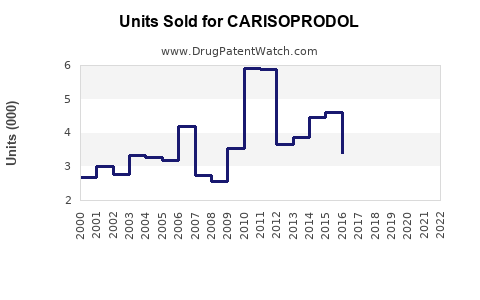
- In 2020, US sales of carisoprodol approximate $80 million, derived mainly from outpatient prescriptions.
- Global sales are estimated at $120 million, considering regional variations and limited use in certain countries.
- Sales declined by approximately 15% from 2015 to 2020, correlating with increased awareness of dependency risks and regulatory restrictions.
Key Market Drivers
- Clinical Utility: Widely prescribed for acute musculoskeletal conditions requiring muscle relaxation.
- Physician Prescribing Patterns: Preference varies based on safety profiles; decline in usage due to dependency concerns.
- Regulatory Environment: Stricter controls inhibit widespread use, reducing market size.
- Alternative Therapies: Increased adoption of non-scheduled muscle relaxants and physical therapies diminishes demand.
Market Challenges
- Regulatory Restrictions: Scheduled status limits prescription volume; heightened scrutiny impacts sales.
- Safety Concerns: Growing awareness of dependence potential dissuades prescribers.
- Limited Innovation: No recent formulation or patent extensions; market saturation occurs.
- Legal Risks: Prescribing limitations increase liability and reduce prescribing incentives.
Future Sales Projections (2023–2027)
| Year |
US Market Sales (million USD) |
Global Market Sales (million USD) |
| 2023 |
65 |
90 |
| 2024 |
60 |
85 |
| 2025 |
55 |
80 |
| 2026 |
50 |
75 |
| 2027 |
45 |
70 |
Assumptions: Continued regulatory restrictions and safety concerns will suppress sales. The US market may stabilize with fluctuations due to prescriber hesitancy; international markets may experience minor growth aligning with regulatory shifts.
Comparison with Other Muscle Relaxants
- Cyclobenzaprine: US sales exceeded $300 million in 2020, nearly four times that of carisoprodol, due to broader approval and minimal Schedule restrictions.
- Methocarbamol: Sales in the same period were approximately $100 million, offering competition with different regulatory profiles.
- Tizanidine: Gains market share owing to its safer profile and scheduling, further pressuring carisoprodol sales.
Strategic Considerations
- Manufacturers must adapt to increased regulatory oversight by exploring reformulation or novel delivery methods.
- The market potential remains limited unless regulatory relaxations occur or new therapeutic indications are identified.
- Investment in alternative muscle relaxants with safer profiles becomes more attractive.
Key Takeaways
- Global sales of carisoprodol have declined due to safety concerns and regulatory restrictions.
- The drug generates approximately $120 million annually, with US sales around $80 million.
- Future growth is unlikely; sales are projected to decrease steadily through 2027.
- Competition from newer, less restrictive muscle relaxants limits market expansion.
- Regulatory environment and safety profile are primary factors influencing market outlook.
FAQs
1. Why has the market for carisoprodol declined?
The decline results from increased awareness of dependency risks, regulatory restrictions (Schedule IV in the US), and competition from safer alternatives.
2. Are there upcoming regulatory changes that could revive the market?
No major regulatory relaxations are currently planned; future changes depend on clinical re-evaluation or new therapeutic evidence.
3. How does safety profile influence market share?
Safer alternatives with lower dependence potential are more attractive for prescribers, limiting carisoprodol’s share.
4. What are the key international differences affecting sales?
Some countries restrict or have tighter controls on carisoprodol, reducing prescriptions compared to the US market.
5. Will new formulations or patents affect future sales?
Unlikely, as there have been no recent patent extensions or reformulations aimed at market expansion, and safety concerns remain dominant.
References
[1] IQVIA. (2020). Pharmaceutical Market Data.
[2] FDA. (2012). Boxed Warning and Schedule IV Status.
[3] DEA. (2012). Scheduling Actions.
[4] Global Data. (2021). Muscle Relaxant Market Reports.
[5] Statista. (2021). US Drug Sales Data.