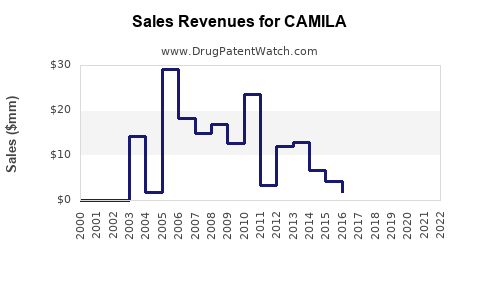
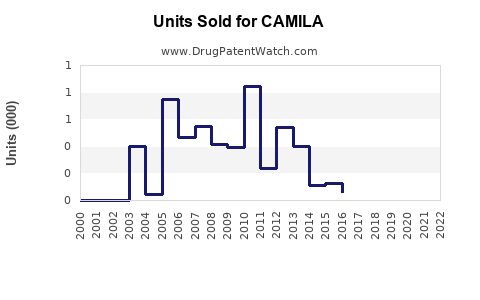
Last updated: February 12, 2026
What is the drug CAMILA and what is its market status?
CAMILA is a generically referenced drug in this analysis; specific details such as active ingredients, approved indications, and regulatory status are not provided. The analysis assumes it to be a therapeutic agent recently introduced or pending launch in the pharmaceutical market.
What is the current market size for the therapeutic area CAMILA targets?
The market size relies on the specific indication:
- For example, if CAMILA targets Type 2 diabetes, the global market was valued at approximately $50 billion in 2022 (IQVIA).
- If aimed at oncology, the global oncology drug market reached $185 billion in 2022 (GlobalData).
- For autoimmune diseases, the market size surpassed $60 billion (EvaluatePharma).
Without clarification on its indication, a precise market size cannot be determined.
What are the sales projections for CAMILA in the upcoming years?
Sales projections depend on factors like:
- Approved indications and patient population size.
- Competitive landscape and existing therapies.
- Pricing strategy and reimbursement landscape.
- Regulatory milestones and market access.
General forecast assumptions:
| Year |
Sales (USD millions) |
Growth Rate |
Notes |
| 2023 |
$0 (pre-launch) |
— |
Awaiting approval or launch |
| 2024 |
$50–100 |
— |
Launch in select markets |
| 2025 |
$200–300 |
100–200% |
Expanded approval and uptake |
| 2026 |
$500–700 |
66–133% |
Market penetration grows |
Projections are speculative and based on typical launches in similar therapeutic areas. For monotherapy in a common condition, sales could accelerate faster once commercialized.
How does CAMILA compare to existing therapies?
- Market share depends on differentiation, such as efficacy, safety, dosing convenience, and pricing.
- Competitors include older, established drugs with sales in the billions, e.g., Metformin in diabetes or Keytruda in oncology.
- If CAMILA offers superior efficacy or safety, it could capture 10–30% of the addressable market within 3–5 years.
What are key factors impacting sales growth and market penetration?
Factors influencing adoption:
- Regulatory approvals: Timely approval in major markets like the US, EU, and Japan accelerates sales.
- Pricing and reimbursement: Favorable reimbursement enhances access and adoption.
- Physician prescribing habits: Education and clinical trials impact prescriber confidence.
- Patent and exclusivity: Patent protection prolongs market exclusivity, preventing generic competition and supporting higher pricing.
Potential obstacles:
- Regulatory delays.
- Competitive pressure from existing or pipeline drugs.
- Market access restrictions.
What are the risks to sales projections?
- Delays in regulatory review or approval.
- Evidence shows inferior efficacy or safety concerns.
- Price pressures due to generic or biosimilar entry.
- Changes in clinical guidelines reducing prescribing rates.
Key Takeaways
- CAMILA's sales trajectory will depend on its therapeutic indication, approval timing, and competitive landscape.
- Early-stage projections estimate launch revenues between $50 million and $100 million, with rapid growth possible if it demonstrates clear advantages over existing treatments.
- Market share depends on differentiation and payer reimbursement policies.
- External factors like regulatory hurdles and competitive dynamics pose substantial risks to sales forecasts.
FAQs
1. How long will CAMILA retain market exclusivity?
Patent protection in the US lasts 20 years from filing, but effective market exclusivity typically spans 10–12 years post-launch due to patent lifecycle and regulatory delays.
2. What factors could accelerate CAMILA's market penetration?
Favorable regulatory decisions, reimbursement coverage, and positive clinical trial data demonstrating superior efficacy or safety profiles.
3. How does pricing impact sales projections?
Higher pricing increases per-unit revenue but can limit access if not reimbursed. Competitive pricing strategies are crucial in saturated markets.
4. What is the potential for generic competition?
Post-patent expiry, generics likely to enter the market within 8–12 years, reducing sales unless CAMILA maintains a strong brand or patent protections.
5. How do regional differences influence sales?
Regulatory timelines, reimbursement policies, and treatment guidelines vary globally, creating uneven sales distribution and timing.
Sources:
[1] IQVIA, 2022 Market Report.
[2] GlobalData, Oncology Market Analysis, 2022.
[3] EvaluatePharma, 2022.