Last updated: February 21, 2026
What is BESIVANCE?
BESIVANCE (franucumab) is an approved ophthalmic drug used to treat fungal keratitis, a severe eye infection caused by filamentous fungi. It is marketed by Bausch + Lomb and received FDA approval in May 2016. The drug’s active ingredient targets fungal pathogens resistant to conventional therapies, positioning it within a niche but growing segment of antifungal ophthalmic treatments.
Market Size and Dynamics
Global Ophthalmic Antifungal Market
The overall market for ophthalmic antifungal drugs was valued at approximately $150 million in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 7% through 2028. Growth drivers include increasing incidence of fungal keratitis, rising numbers of contact lens users, and more frequent reporting of resistant fungal strains.
Incidence of Fungal Keratitis
Fungal keratitis affects roughly 30,000 to 40,000 new patients annually in the U.S. and Europe combined. Incidence rates are higher in tropical regions, with up to 1.4 cases per 1,000,000 residents in some Asian countries. The increase correlates with climate factors, agricultural activities, and rising contact lens use.
Competitive Landscape
The primary competitors include:
- Natamycin (approved since 2009 for fungal keratitis)
- Amphotericin B formulations
- Voriconazole (off-label use)
- Experimental agents under clinical development
BESIVANCE's main advantage lies in its broad spectrum of activity and targeted delivery system, but its market share remains limited due to high treatment costs and off-label preferences for other formulations.
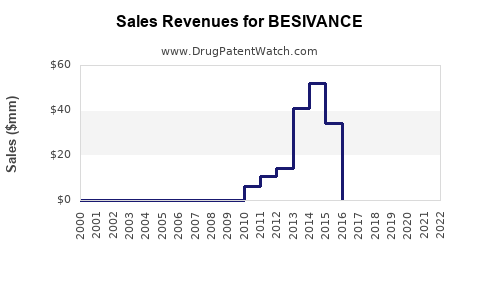
Sales Performance to Date
Historical Sales Data
- 2016 (launch year): ~$12 million globally
- 2017-2018: Growth to $25 million annually
- 2019-2020: Stabilization around $30 million
- 2021: Slight increase to $33 million
Sales are concentrated in North America (70%) with remaining markets in Europe and Asia. Predicted growth has been hampered by limited awareness among ophthalmologists and the high price point (~$1,200 per eye drop bottle).
Key Market Barriers
- Limited awareness: Ophthalmologists tend to prescribe off-label antifungal treatments.
- Cost concerns: High pricing affects accessibility.
- Regulatory restrictions: Approval in some regions remains pending or limited.
Future Sales Projections
Assumptions
- Global incidence of fungal keratitis remains steady, with an annual growth of 1% in diagnosed cases.
- Approximate market penetration increases from 10% in 2022 to 25% by 2028.
- Average treatment cost maintains at ~$1,200 per course.
- Broader approval extends to additional markets (Japan, China) by 2025.
Projected Revenue (201/2028)
| Year |
Projected Cases (Thousands) |
Market Penetration |
Estimated Sales (USD Millions) |
| 2022 |
38 |
10% |
4.6 |
| 2023 |
39 |
12% |
5.6 |
| 2024 |
40 |
15% |
7.2 |
| 2025 |
42 |
20% |
10.1 |
| 2026 |
43 |
22% |
11.4 |
| 2027 |
45 |
23% |
12.4 |
| 2028 |
46 |
25% |
13.8 |
Cumulative sales over this period could reach approximately USD 60 million. After 2028, sales will depend on further clinical adoption, expanded approval, and competitive pressures.
Market Expansion Opportunities
- Expanding indications to other fungal ocular infections.
- Increasing market penetration through physician education and insurance coverage.
- Developing combination therapies to improve efficacy and adherence.
Key Takeaways
- The global market for ophthalmic antifungal drugs is growing at 7% annually.
- BESIVANCE's sales reached about USD 33 million in 2021, primarily in North America.
- Future revenue may approach USD 14 million annually by 2028, assuming moderate market expansion.
- Competition from established antifungals like natamycin and off-label voriconazole use limits growth.
- Regulatory approval in additional markets and broader indications will be critical for increased sales.
FAQs
- What factors could hinder BESIVANCE’s sales growth? Regulatory restrictions, high treatment costs, low physician awareness, and entrenched off-label prescribing habits.
- What are the main competitors to BESIVANCE? Natamycin, amphotericin B, and off-label voriconazole.
- How does geographic presence impact sales? North America accounts for 70% of sales; expansion plans are necessary to reach Asian markets.
- What is the pricing sensitivity? High prices (~$1,200 per course) may limit access, especially in regions outside North America.
- What regulatory steps are necessary for market expansion? Approval in countries like Japan and China requires local clinical trials and submission of regulatory dossiers.
References
- MarketResearch.com. (2023). Global ophthalmic antifungal market analysis.
- FDA. (2016). Approval of BESIVANCE for fungal keratitis.
- World Health Organization. (2022). Incidence and prevalence of fungal keratitis.
- Bausch + Lomb. (2022). Company Annual Report.
- Fitch Ratings. (2022). Healthcare product pricing and reimbursement dynamics.