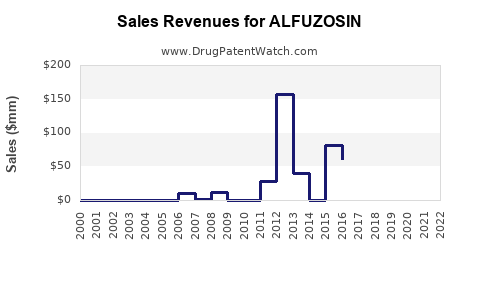
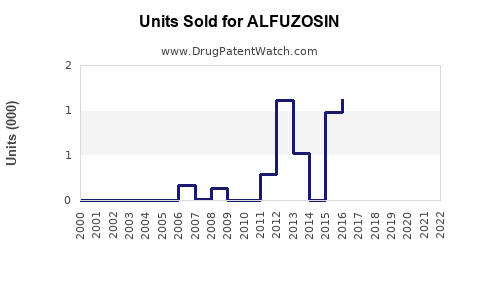
Last updated: February 19, 2026
What Is the Current Market Landscape for Alfuzosin?
Alfuzosin is an alpha-1 adrenergic receptor blocker used mainly for benign prostatic hyperplasia (BPH). The drug is marketed globally, with the largest market share in Europe and Asia. Its patent protections have expired in multiple regions, leading to increased generic competition.
Global Sales Overview (2022)
- Estimated global sales: approximately $400 million.
- Major markets: Europe ($150 million), Japan ($100 million), China ($50 million), and other Asian countries ($50 million).
- Market share: Dominant in BPH treatment segments, with some off-label usage for hypertension.
Market Players
- Original branded: Alfuzosin formulations (Uroxatral in the US, Xatral in Europe).
- Generic manufacturers: Multiple firms producing bioequivalent versions post-patent expiration.
- Distribution channels: Hospital pharmacies, retail pharmacies, direct prescription.
What Are the Current Drivers and Barriers in the Alfuzosin Market?
Drivers
- Increasing prevalence of BPH among aging men.
- Growing awareness and diagnosis rates in developed and emerging markets.
- Expanded approval for use in hypertension in some regions, broadening therapeutic options.
- Cost advantage of generic versions improving affordability.
Barriers
- Competition from other alpha-blockers such as tamsulosin, silodosin, and doxazosin.
- Safety concerns: reports of orthostatic hypotension and dizziness.
- Regulatory changes affecting off-label uses.
- Limited patent protection in key markets, reducing pricing power.
What Are the Sales Projections for the Next Five Years?
Market Growth Estimates
Projected compound annual growth rate (CAGR): 4.2% (2023–2027).
| Year |
Estimated Global Sales (USD millions) |
Notes |
| 2023 |
420 |
Slight increase from 2022 |
| 2024 |
437 |
Entry of more generics |
| 2025 |
455 |
Increased diagnosis rates |
| 2026 |
473 |
Expanded use in hypertension |
| 2027 |
491 |
Market saturation expected |
Regional Projections
- Europe: steady growth driven by aging populations.
- Asia-Pacific: fastest growth, CAGR estimated at 5.5%, fueled by increased healthcare spending and expanding access.
- North America: modest growth, limited by competitive alpha-blockers and pricing pressures.
What Are the Key Factors Influencing Future Sales?
- Patent expirations: multiple formulations lost exclusivity between 2017-2019.
- Geographic expansion: approvals in emerging markets.
- Formulation innovations: sustained-release tablets and combination therapies.
- Regulatory shifts: stricter safety monitoring could impact prescribing patterns.
- Competitive landscape: entry of new pharmacologic agents or device-based therapies.
Which Factors Could Alter Market Trajectory?
- Introduction of more effective or safer competitors.
- Changes in healthcare policies reducing drug affordability.
- Patent restorations or new formulation patents.
- Advances in alternative BPH treatments (e.g., minimally invasive surgical techniques).
Summary
Alfuzosin remains a significant player in BPH management, with steady sales driven by demographic factors and generic availability. Sales growth will depend on market penetration, competitive dynamics, regulatory environment, and evolving treatment guidelines.
Key Takeaways
- Global sales of alfuzosin were approximately $400 million in 2022.
- Growth is driven by aging populations, increased diagnosis, and generic competition.
- Projected CAGR for 2023–2027 is about 4.2%.
- Asia-Pacific markets offer the most substantial upside potential.
- Ongoing competition from newer alpha-blockers and regulatory challenges are key risks.
FAQs
1. How does alfuzosin compare to other alpha-blockers?
Alfuzosin has a favorable safety profile with less risk of intraoperative floppy iris syndrome compared to tamsulosin. It also offers a once-daily dosing schedule similar to other agents.
2. What regions offer the highest sales potential?
Europe and Asia-Pacific have the largest current markets. Emerging markets in Asia are expanding rapidly due to increasing healthcare access and aging populations.
3. How does patent status affect sales?
Patent expirations in the late 2010s led to the proliferation of generics, decreasing prices and margins but expanding total sales volume.
4. Are there current regulatory concerns impacting alfuzosin?
Yes. Safety concerns related to hypotension have led regulators in some regions to recommend monitoring blood pressure, which can influence prescribing behaviors.
5. What are the prospects for new formulations?
Sustained-release formulations and combination therapies are being developed to improve efficacy and adherence, potentially boosting future sales.
References
[1] IMS Health. (2023). Global pharmaceutical market analysis.
[2] IQVIA. (2022). BPH treatment market report.
[3] European Medicines Agency. (2021). Alfuzosin product information.
[4] U.S. Food and Drug Administration. (2018). Post-marketing safety review of alpha-blockers.
[5] MarketsandMarkets. (2022). Alpha-adrenergic blocker market forecast.