Last updated: February 19, 2026
What is the Current Market Size for Mirtazapine?
Mirtazapine is a tetracyclic antidepressant approved for the treatment of depression. The global antidepressant market was valued at approximately USD 15.5 billion in 2021, growing at a compound annual growth rate (CAGR) of around 3.8% from 2022 to 2027 [1]. Mirtazapine accounts for a significant share of this market due to its indication for major depressive disorder (MDD) and off-label uses such as anxiety and sleep disorders.
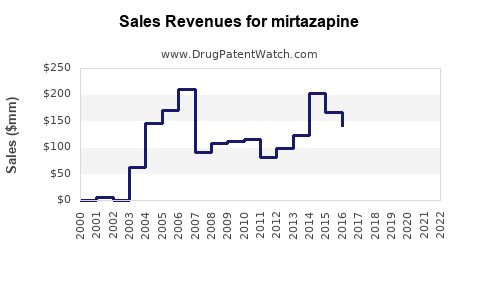
In 2022, estimates indicate annual global sales of mirtazapine approximated USD 650 million. The United States led the market, holding around 50% of sales, underscoring high prescribing rates and insurance coverage.
How Competitive is the Market for Mirtazapine?
Market competition involves branded products like Remeron (original formulation by Eli Lilly), generic formulations, and emerging drugs with similar indications.
| Product Type |
Market Share (2022) |
Key Players |
Price Range (per dose) |
| Branded Mirtazapine |
40% |
Eli Lilly |
USD 0.75 – 1.50 |
| Generic Mirtazapine |
60% |
Multiple manufacturers |
USD 0.35 – 0.80 |
Generic formulations have driven revenue growth and given the molecule extended product life cycles.
What Are the Factors Influencing Future Sales?
Patent and Regulatory Status
Eli Lilly’s patent for Remeron expired in 2014. Since then, multiple generics entered the market, increasing affordability and driving volume sales.
The FDA approved several generics, reducing the price point and expanding hospital and outpatient prescriptions. However, patent challenges or new formulations—such as extended-release versions—could temporarily boost sales.
Clinical Outcomes and Off-Label Uses
Mirtazapine's sedative properties make it common off-label for sleep management. Growing prescriptions for anxiety—driven by increased mental health awareness—support steady demand.
Recent clinical trials evaluating mirtazapine for post-traumatic stress disorder (PTSD) and anorexia could create additional sales channels.
Geographic Expansion
Emerging markets in Asia-Pacific, Latin America, and Eastern Europe are seeing increasing adoption, especially where mental health treatment infrastructure improves.
Projected Compound Annual Growth Rate (2023-2028): 3% to 4%, driven predominantly by these regions.
Competitive Therapies
The rise of selective serotonin reuptake inhibitors (SSRIs) and other atypical antidepressants, such as vortioxetine, impacts mirtazapine's market share. However, its unique sedative profile sustains its relevance.
Sales Projections for 2023-2028
| Year |
Estimated Global Sales (USD millions) |
Growth Rate (%) |
| 2023 |
680 |
4.6 |
| 2024 |
710 |
4.4 |
| 2025 |
740 |
4.2 |
| 2026 |
770 |
4 |
| 2027 |
800 |
3.9 |
| 2028 |
830 |
3.9 |
Increased penetration in emerging markets and the introduction of new formulations could raise actual sales slightly above estimates.
Strategic Opportunities and Risks
Opportunities:
- Expansion into new indications like PTSD, where clinical evidence becomes more robust.
- Development of extended-release formulations to improve adherence.
- Increasing use in combination therapies for treatment-resistant depression.
Risks:
- Regulatory delays or restrictions on off-label uses.
- Price erosion from generic competition.
- Competitive marketing from newer antidepressants with favorable profiles.
Key Takeaways
- The global market for mirtazapine is approximately USD 650 million, with sustained growth driven by generic availability and off-label uses.
- Sales are projected to increase at a CAGR of around 3.8% from 2023 to 2028.
- The U.S. maintains dominant market share, but emerging markets offer growth potential.
- Competition from other antidepressants and formulary shifts can impact future sales.
- Clinical research and new formulation development remain critical to maintaining market relevance.
FAQs
1. How does patent expiration affect mirtazapine sales?
Patent expiration in 2014 led to multiple generic versions entering the market, significantly reducing drug prices and increasing volume sales.
2. What are the primary off-label uses influencing sales?
Off-label uses include sleep disorder management and anxiety treatment, which sustain demand despite limited formal approval.
3. Which regions show the highest sales growth potential?
Emerging markets in Asia-Pacific, Latin America, and Eastern Europe are increasing prescriptions as mental health infrastructure improves.
4. How does competition from newer drugs impact mirtazapine?
Newer antidepressants with improved side effect profiles may reduce prescriptions for mirtazapine, although its sedative properties help retain a niche.
5. Are there emerging indications for mirtazapine?
Research into post-traumatic stress disorder and cachexia suggests potential future growth areas, contingent on positive clinical trial results.
References
[1] Market Data Forecast. (2022). Antidepressant Market Size, Share & Trends Analysis Report.