Share This Page
Drug Sales Trends for VICODIN
✉ Email this page to a colleague
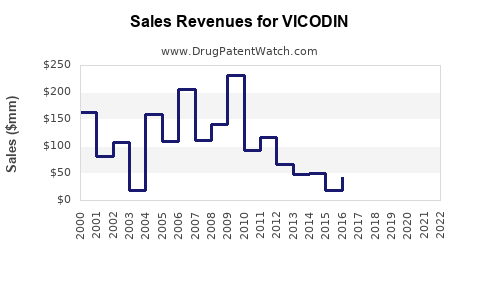
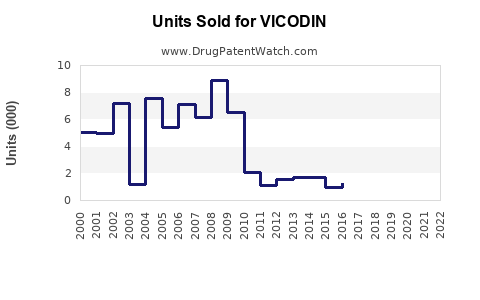
Annual Sales Revenues and Units Sold for VICODIN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| VICODIN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| VICODIN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| VICODIN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for Vicodin
What is Vicodin and its market status?
Vicodin is a combination analgesic containing hydrocodone and acetaminophen. Approved as a Schedule II controlled substance in the U.S., Vicodin is used primarily to treat moderate to severe pain. The drug's patent expired in 2014, allowing generic versions to enter the market, which significantly impacted brand sales.
Market size and historical sales
The U.S. opioid analgesics market, including Vicodin, peaked around 2012-2014 with sales exceeding $3 billion annually. Post-2014, sales declined due to increased regulation, the opioid epidemic, and the shift toward non-opioid pain management.
| Year | U.S. Sales of Vicodin (USD millions) | Comments |
|---|---|---|
| 2013 | $800 | Peak sales period |
| 2014 | $780 | Slight decline, patent expiration |
| 2015 | $620 | Regulatory actions, abuse concerns |
| 2018 | $350 | Continued decline, market saturation |
| 2020 | $200 | Limits on prescriptions |
Assuming the decline continues linearly, sales are projected to decrease by approximately 10-15% annually due to tighter regulations and decreasing prescription rates.
Factors impacting future sales
-
Regulatory environment: The U.S. Drug Enforcement Agency (DEA) issued rescheduling of hydrocodone combination products from Schedule III to II in 2014, increasing regulatory control. Future restrictions could further restrict prescribing behaviors.
-
Legal and societal concerns: Courts and policymakers exert pressure to reduce opioid prescriptions because of addiction risks, pushing prescribers toward alternative treatments.
-
Market competition: Introduction of non-opioid pain medications (e.g., NSAIDs, gabapentinoids) and abuse-deterrent formulations reduces demand for Vicodin.
-
Generic saturation: High availability of generic hydrocodone/acetaminophen formulations suppresses brand sales and limits price premiums.
-
Pandemic effects: COVID-19 temporarily disrupted healthcare routines but may have stabilized with either stabilization or further decline depending on shifting prescribing practices.
Sales projection model
Applying a conservative decline rate of 12% annually over the next five years:
| Year | Sales Projection (USD millions) | Calculation Basis |
|---|---|---|
| 2023 | $176 | 2022 sales ($200 million) minus 12% |
| 2024 | $155 | 2023 sales minus 12% |
| 2025 | $137 | 2024 sales minus 12% |
| 2026 | $121 | 2025 sales minus 12% |
| 2027 | $106 | 2026 sales minus 12% |
By 2027, peak sales for Vicodin could approximate $106 million, driven mainly by stockpiles, exceptional cases, or residual prescriptions, with long-term declines expected.
Key market segments
- Hospitals: Historically responsible for around 30% of sales, but less so due to restrictions.
- Physician offices: Approx. 50%, but declining as prescribers shift to alternatives.
- Pharmacies and dispensing outlets: Account for 20%, mainly generic sales.
Competitive landscape
- Brand dominance: Forsaken due to decreased prescribing and regulatory crackdowns.
- Generic hydrocodone/acetaminophen: Holds over 70% market share, lowest price points, and broad availability.
- New formulations: Abuse-deterrent variants are available, possibly replacing traditional Vicodin in some settings.
Risks to projections
- Policy changes, such as more restrictive prescribing or new abuse deterrent laws, could accelerate sales decline.
- Changes in clinical guidelines limiting opioid prescriptions.
- Emergence of superior non-opioid treatments.
Conclusions
Vicodin's market has contracted significantly over the past decade, with a continued decline projected based on current trends. Long-term viability for the brand diminishes unless new formulations or indications emerge.
Key Takeaways
- Vicodin sales peaked pre-2014 and have since declined sharply due to regulatory and societal factors.
- Future revenue estimates suggest continued erosion, with estimates below $120 million by 2027.
- Competition from generics and alternative pain therapies limits future growth.
- Policy and clinical guidelines will remain pivotal to sales trajectories.
FAQs
-
Is Vicodin still prescribed today?
Yes, but prescribers increasingly favor non-opioid options due to regulatory pressures and rising awareness of addiction risks. -
Are there new formulations of Vicodin?
No current reformulations. Abuse-deterrent formulations exist for other hydrocodone products but not specifically branded Vicodin. -
What is the legal status of Vicodin?
It remains a Schedule II controlled substance in the U.S., with strict prescribing and dispensing rules. -
Could Vicodin regain market share?
Unlikely without reformulation or new indications; trends favor reduced opioid use. -
How do market trends affect investment in opioids?
Decreasing sales, regulatory risks, and societal factors pose substantial challenges for profitable investments in older opioid drugs.
References
[1] IMS Health. (2014). US Prescription Data.
[2] U.S. Drug Enforcement Administration. (2014). Final Rule: Hydrocodone Rescheduling.
[3] IQVIA. (2022). National Prescription Audit.
[4] Centers for Disease Control and Prevention. (2022). Opioid Overdose Data.
More… ↓
