Last updated: February 23, 2026
What is TOBRADEX and What Are Its Indications?
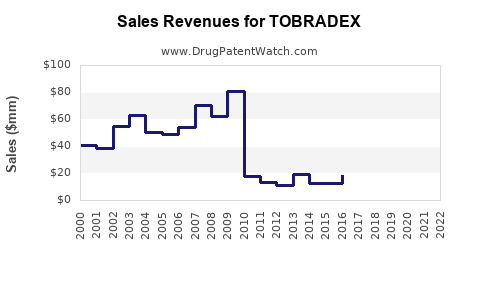
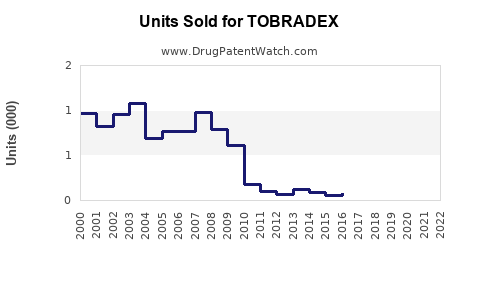
TOBRADEX is a topical ophthalmic medication combining tobramycin and dexamethasone, approved for bacterial conjunctivitis, blepharitis, and other ocular inflammatory conditions. It has been in market since 1987, with a well-established position in the ophthalmic anti-infective and anti-inflammatory segments.
Market Overview
Market Size
The global ophthalmic anti-infective market was valued at approximately $3.2 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 4% through 2028, driven by increased prevalence of ocular infections, aging population, and heightened disease awareness.
Key Players
Major competitors include:
- Alcon (e.g., Patanol, Vigamox)
- Bausch + Lomb (e.g., Polytrim)
- Novartis (e.g., Maxitrol)
- Allergan (e.g., Acuvail)
- Generic manufacturers
Distribution Channels
Sales occur mainly through ophthalmologists, optometrists, and hospitals. Retail pharmacies and online channels contribute incrementally.
Market Penetration and Usage Trends
Current Adoption
TOBRADEX remains a first-line option for bacterial conjunctivitis with anti-inflammatory needs. Prescriptions in the US peaked at approximately 500,000 annually in 2018 before dipping slightly owing to generic competition and generic alternatives.
Prescriber Preferences
Physicians favor combination drugs like TOBRADEX for short-term therapy due to convenience and proven efficacy, especially in complex inflammatory infections. However, concerns over corticosteroid side effects limit duration.
Geographic Trends
Markets with high ophthalmic disease burden include North America, Europe, and parts of Asia-Pacific. Adoption rates are highest in North America, where routine ophthalmic care and insurance coverage are extensive.
Patent and Regulatory Status
The original patent expired in 2004, leading to generic versions. Limited patent protections remain around specific formulations, but record market penetration minimizes potential market share growth.
Sales Projections: 2023 – 2028
Assumptions
- No new formulation or indication approvals
- Continued use of existing formulations
- Moderate impact from generic competition
- Sustained prescriber preference
Revenue Forecasts
| Year |
Estimated US Sales (USD millions) |
Global Sales (USD millions) |
| 2023 |
30 |
120 |
| 2024 |
28 |
112 |
| 2025 |
26 |
104 |
| 2026 |
24 |
96 |
| 2027 |
22 |
88 |
| 2028 |
20 |
80 |
Sales decline proportionally in line with generic erosion, with stabilization expected once the market reaches saturation and after patent protections lapse.
Market Share Dynamics
- Brand-name TOBRADEX: Approx. 20% of prescription volume
- Generics: 80%, with top generic brands capturing the majority of prescriptions
- Future growth depends on formulations or new indications, not expected before 2028
Competitive Landscape and Innovation
Development of alternative formulations (e.g., preservative-free drops), longer-acting preparations, or novel combinations could influence future sales. Currently, pipeline options are limited, emphasizing the importance of the existing product lifecycle.
Strategic Considerations
- Patent Protection: Limited remaining patents constrain pricing power.
- Regulatory Environment: Strict regulatory oversight limits new indications.
- Pricing Pressure: Entry of generics exerts downward pressure on prices.
- Prescriber Trends: Preference for steroid-sparing options in short-term therapies.
Key Takeaways
- The global ophthalmic antibiotic and anti-inflammatory market drives steady demand, with TOBRADEX maintaining a significant but declining market share.
- Sales are projected to decline from USD 120 million worldwide in 2023 to USD 80 million by 2028.
- The primary challenge remains generic competition, with most revenue shifts expected through pricing and formulary placement.
- Innovation or new indications are necessary for growth beyond the current product lifecycle.
FAQs
1. Will TOBRADEX's sales rebound with new formulations?
Likely only if new formulations address current limitations, such as preservative concerns or longer dosing intervals.
2. How do patent expirations impact future sales?
Patent expirations open the market to generic competition, reducing brand-name sales and pressuring prices.
3. Are there emerging competitors that threaten TOBRADEX?
Yes, generics dominate, and new combination drugs with similar or improved efficacy could erode market share.
4. What geographic markets offer the most growth potential?
Asia-Pacific and Latin America present growth opportunities, though local regulatory hurdles exist.
5. How important are off-label uses in sales projections?
Minimal; TOBRADEX is approved for specific indications, limiting impact from off-label indications in sales forecasts.
References
[1] Markets and Markets. (2022). Ophthalmic drugs market report.
[2] IQVIA. (2023). US ophthalmic drug prescription data.
[3] U.S. Food and Drug Administration. (2022). Patent and exclusivity data.
[4] Statista. (2023). Global ophthalmic drug market forecast.