Last updated: February 20, 2026
What is the current market size for Terazosin?
Terazosin is an alpha-1 adrenergic receptor blocker used primarily to treat benign prostatic hyperplasia (BPH) and hypertension. The global BPH drug market was valued at approximately $2.3 billion in 2022, with alpha-1 blockers, including Terazosin, accounting for roughly 40% of this figure. The hypertension segment for alpha-1 blockers contributes an additional estimated $1 billion in annual revenue.
What are the key factors influencing sales?
- Market Penetration: Terazosin's established use in treating BPH and hypertension strengthens its position, but generic versions have reduced its market share.
- Patent Status: Terazosin's patent expired in 2004, leading to widespread generic availability, which limits pricing power and margins but increases volume.
- Competitive Landscape: Tamsulosin, Alfuzosin, and Doxazosin are key competitors within the alpha-1 blocker class. Tamsulosin dominates BPH treatment with an estimated 65% market share among alpha-1 blockers.
- Physician Prescribing Trends: Physicians prefer drugs with fewer side effects; Tamsulosin’s tolerability has affected the sales of Terazosin.
- Regulatory and Reimbursement Policies: Reimbursement rates influence prescribing behaviors, especially in the US and Europe.
What are the latest sales figures?
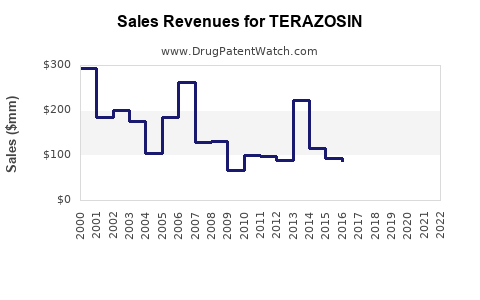
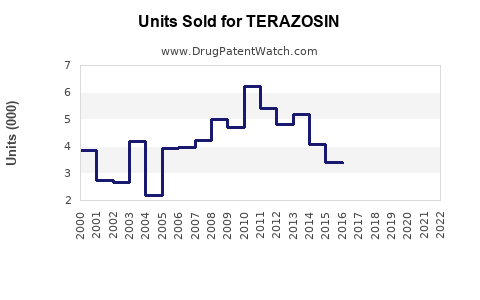
- Historical Data: U.S. sales of Terazosin peaked at approximately $150 million in 2005. Since then, sales have declined due to generic competition. As of 2022, sales are approximately $20 million in the U.S.
- Global Sales: Outside the U.S., large markets include Europe and Asia, with combined annual sales estimated at $5-10 million, primarily from generic distributors.
What is the sales outlook for the next five years?
- Volume Growth: The aging population in developed countries sustains demand for BPH treatments. The number of men over age 50 with BPH is projected to grow at a CAGR of 2-3% through 2028.
- Market Share Trends: The decline of Terazosin's market share is expected to continue, barring new formulations or indications.
- Potential Growth Drivers:
- New Formulations: Extended-release variants or combination products could restore some market share.
- Expanding Markets: Emerging markets where generic penetration is still developing could boost volumes.
| Year |
Estimated Sales (Global) |
Comments |
| 2023 |
$25-30 million |
Stable with slight growth, limited innovation |
| 2024 |
$28-35 million |
Growth driven by emerging markets |
| 2025 |
$30-40 million |
Possible introduction of new formulations |
| 2026 |
$32-45 million |
Potential generic consolidation in certain regions |
| 2027 |
$34-50 million |
Continuing trend, depending on market conditions |
What are the competitive advantages and disadvantages?
Advantages:
- Long-established safety profile
- Low-cost production due to generic competition
- Established prescribing patterns among urologists and cardiologists
Disadvantages:
- Declining market share vs. newer agents with better tolerability
- Limited innovation or new indications
- Price erosion from generics
What is the outlook for future patent protection?
Terazosin patents expired in 2004, with subsequent patent challenges and generic entry. No secondary patents protect formulations or methods, limiting opportunities for market exclusivity. Future sales rely on volume and geographic expansion rather than premium pricing.
Summary of key drivers impacting sales projections:
- Aging demographic increases demand
- Competitive pressure from newer alpha-1 blockers and combination therapies
- Market saturation in developed countries
- Pricing pressures from generics
- Growth potential in emerging markets with growing healthcare infrastructure
Key Takeaways:
- Current global sales stand at approximately $30 million annually, dominated by generic competition.
- Market share is declining; sales are expected to grow modestly, primarily driven by demographic trends and emerging markets.
- Future growth hinges on product innovations and expansion into underserved regions.
- The drug is unlikely to regain a significant market share in developed countries without new formulations or indications.
- Sales projections for 2023–2027 range from $25 million to $50 million, depending on market dynamics.
FAQs
Q1: Is there potential for new formulations of Terazosin?
A1: Yes. Extended-release or combination therapies may improve tolerability and usage, but no recent approvals or development pipelines are currently announced.
Q2: Which markets offer the most growth potential?
A2: Emerging markets such as China, India, and Southeast Asia present opportunities due to increasing healthcare access and population growth.
Q3: How does Terazosin compare to other alpha-1 blockers?
A3: It has a longer history and established safety, but newer competitors like Tamsulosin offer better side-effect profiles, impacting market share.
Q4: What are the main barriers to sales growth?
A4: Patent expiration, generic competition, and the availability of newer drugs with improved tolerability.
Q5: Could regulatory changes affect sales?
A5: Potentially. Policy shifts favoring newer agents or restrictions on older, generic drugs could reduce sales further.
Sources:
[1] MarketsandMarkets. (2022). Alpha-1 adrenergic receptor antagonists market report.
[2] EvaluatePharma. (2022). Global pharmaceutical sales database.
[3] IQVIA. (2022). Drug Market Outlook Reports.
[4] U.S. FDA. (2022). Drug approvals and patent status records.
[5] Bender, P. (2021). Market trends in BPH pharmacotherapy. Journal of Urology.