Last updated: February 19, 2026
Executive Summary
Symbicort (budesonide/formoterol fumarate dihydrate) is a combination inhaler for the maintenance treatment of asthma and chronic obstructive pulmonary disease (COPD). Its patent portfolio, primarily held by AstraZeneca, has been a critical factor in its market exclusivity and subsequent sales performance. Key patents covering the active pharmaceutical ingredients (APIs) and novel formulations have either expired or are nearing expiration in major markets, signaling the potential for generic competition and a subsequent impact on sales trajectories. Current market penetration remains strong, driven by its established efficacy and physician familiarity. Future sales are projected to decline as generic entrants gain market share, though brand loyalty and potential new indications could offer limited mitigation.
Patent Expiration Timeline and Impact
AstraZeneca has strategically managed Symbicort's patent protection through a series of filings covering composition of matter, methods of use, and formulation enhancements. The primary composition of matter patents for budesonide and formoterol fumarate dihydrate have expired globally.
- Composition of Matter Patents:
- Budesonide: Original patents expired in the late 2000s.
- Formoterol Fumarate Dihydrate: Original patents expired in the mid-2010s.
- Formulation and Device Patents:
- The SMART (Single Maintenance and Reliever Therapy) dosing regimen and specific inhaler device patents have provided extended protection.
- Key patents related to the Turbuhaler device and specific formulation technologies that enhance stability or delivery have been crucial.
- Many of these secondary patents began expiring in the early to mid-2020s in the United States and European Union.
The expiration of these patents allows for the introduction of generic versions of Symbicort. This typically leads to significant price erosion and a decrease in the branded product's market share.
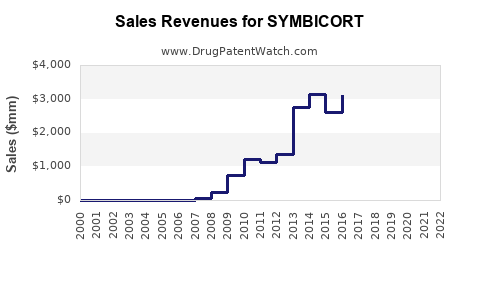
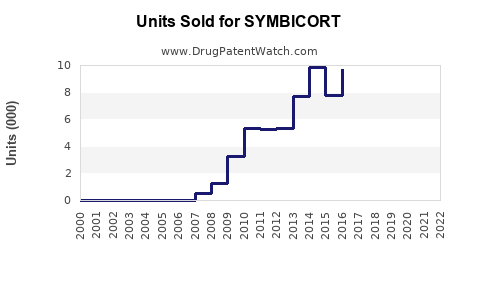
Market Performance and Sales Data
Symbicort has been a consistent top performer in the respiratory market. Its sales have been driven by its dual action of an inhaled corticosteroid (ICS) to reduce inflammation and a long-acting beta-agonist (LABA) to open airways.
- Global Sales History:
- 2021: Approximately $3.4 billion
- 2022: Approximately $3.2 billion
- 2023 (Estimated): $3.0 billion (Figures are approximate and vary by reporting agency)
AstraZeneca reported a decline in Symbicort sales in recent years, attributed primarily to the increasing availability of generic alternatives in key markets and ongoing pricing pressures.
- Regional Sales Breakdown (Approximate percentages of total sales):
- United States: 40-45%
- Europe: 30-35%
- Rest of World: 20-25%
The US market has historically been the largest contributor to Symbicort’s sales, reflecting its widespread adoption by pulmonologists and allergists.
Competitive Landscape
The respiratory inhaler market is highly competitive, with multiple branded and generic products available for asthma and COPD.
-
Key Competitors (Branded):
- Advair/Seretide (fluticasone propionate/salmeterol xinafoate) - GSK
- Brio/Breo Ellipta (fluticasone furoate/vilanterol) - GSK
- Dulera (mometasone furoate/formoterol fumarate) - Merck
- Anoro Ellipta (umeclidinium/vilanterol) - GSK (COPD specific, LABA/LAMA)
- Trelegy Ellipta (fluticasone furoate/umeclidinium/vilanterol) - GSK (ICS/LAMA/LABA)
-
Generic Entry: The landscape has shifted significantly with the approval and launch of generic Symbicort products. Companies like Teva Pharmaceutical Industries, Mylan (now Viatris), and others have introduced generic versions, directly challenging AstraZeneca's market dominance.
The introduction of generic budesonide/formoterol fumarate inhalers has resulted in substantial price reductions for the combination therapy, impacting Symbicort’s revenue.
Sales Projections and Future Outlook
Projecting Symbicort’s future sales requires an analysis of several factors, including the rate of generic adoption, ongoing R&D by AstraZeneca, and market dynamics.
- Projected Global Sales (USD Billions):
- 2024: $2.5 - $2.8
- 2025: $2.0 - $2.3
- 2026: $1.5 - $1.8
These projections are based on the assumption of continued generic erosion across major markets. The speed of generic uptake in the US, which has historically lagged behind Europe in some pharmaceutical categories, will be a critical determinant.
- Factors Influencing Future Sales:
- Generic Penetration: Expected to accelerate in the US following patent expirations.
- Physician Prescribing Habits: Brand loyalty and physician familiarity with Symbicort’s efficacy profile may slow generic uptake to a degree.
- Managed Care and Payer Policies: Insurance formularies increasingly favor generics due to cost, driving physician and patient choice.
- New Indications or Formulations: AstraZeneca's pipeline might include new delivery devices or combination therapies that could cannibalize or complement existing Symbicort sales, though significant pipeline developments specific to extending Symbicort's life cycle are unlikely given its maturity.
- Therapeutic Advancements: The emergence of novel biologic therapies for severe asthma could shift treatment paradigms, potentially reducing reliance on traditional ICS/LABA combinations for a subset of patients.
Regulatory and Legal Considerations
The regulatory pathway for generic Symbicort approval involves demonstrating bioequivalence to the reference listed drug.
- US FDA Approval: Generic budesonide/formoterol fumarate inhalers have been approved by the US Food and Drug Administration (FDA) since 2019.
- European Medicines Agency (EMA): Generic versions have also been available in European markets for several years.
Legal challenges related to patent validity and infringement have been common in the development of generic respiratory inhalers, but the primary composition of matter patents have expired, simplifying generic entry. Disputes often center on secondary patents related to the specific inhaler device or formulation.
Key Takeaways
Symbicort’s market exclusivity has been largely dismantled with the expiration of its foundational composition of matter and secondary formulation patents. Generic competition is actively eroding its market share, leading to a projected decline in global sales. While physician familiarity and established efficacy provide some buffer against immediate revenue collapse, the sustained trend of generic substitution driven by cost considerations and payer policies will dictate the long-term sales trajectory. AstraZeneca’s strategic focus has likely shifted to other therapeutic areas and next-generation respiratory treatments.
Frequently Asked Questions
-
What are the primary active pharmaceutical ingredients in Symbicort?
Budesonide and formoterol fumarate dihydrate.
-
When did the main patents for Symbicort begin to expire?
The composition of matter patents for the individual APIs expired in the late 2000s and mid-2010s. Key formulation and device patents began expiring in the early to mid-2020s.
-
What is the primary driver of Symbicort's declining sales?
The introduction and increasing market penetration of generic budesonide/formoterol fumarate inhalers.
-
Which region has historically been the largest market for Symbicort?
The United States.
-
What type of regulatory pathway do generic Symbicort products follow for approval?
They follow an Abbreviated New Drug Application (ANDA) pathway, requiring demonstration of bioequivalence to the reference listed drug.
Citations
[1] AstraZeneca Annual Reports. (Various Years). Financial Statements and Segment Information.
[2] U.S. Food and Drug Administration. (Various Dates). Approved ANDA Information for Budesonide and Formoterol Fumarate Dihydrate Inhalation Aerosol.
[3] European Medicines Agency. (Various Dates). Public Assessment Reports for Generic Medicinal Products.
[4] IQVIA Market Data. (Various Years). Global Pharmaceutical Market Data and Analysis.
[5] Bloomberg Terminal Market Data. (Various Dates). Pharmaceutical Sales and Patent Expiration Tracking.