Share This Page
Drug Sales Trends for PROTONIX
✉ Email this page to a colleague
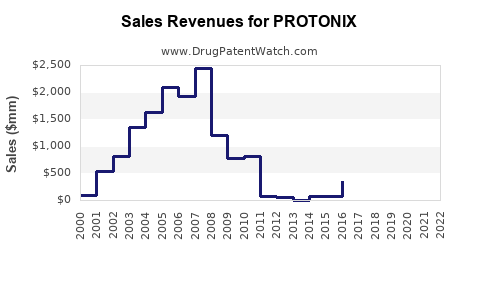
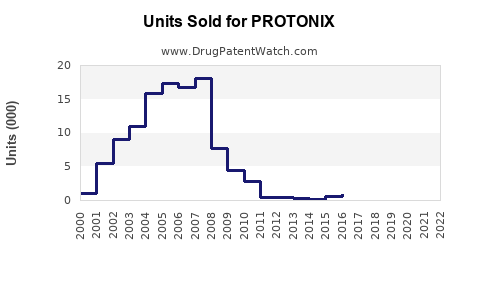
Annual Sales Revenues and Units Sold for PROTONIX
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| PROTONIX | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| PROTONIX | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| PROTONIX | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
PROTONIX (Pantoprazole Sodium) Market Analysis and Sales Projections
Protonix, the brand name for pantoprazole sodium, is a proton pump inhibitor (PPI) used to treat conditions caused by excess stomach acid. Its efficacy in managing gastroesophageal reflux disease (GERD), erosive esophagitis, and Zollinger-Ellison syndrome has established a significant market presence. Patent expirations have led to widespread generic competition, fundamentally altering its market dynamics. This analysis details historical sales, patent status, competitive landscape, and future sales projections for Protonix and its generic equivalents.
What is Protonix and How Does It Function?
Protonix is a benzimidazole derivative that irreversibly blocks the H+/K+-ATPase enzyme system, commonly known as the proton pump, in gastric parietal cells [1]. This action reduces the secretion of gastric acid. It is available in oral formulations (delayed-release tablets and granules) and intravenous (IV) administration. Oral Protonix is typically administered once daily, with dosage varying based on the condition being treated. The IV formulation is used when oral administration is not feasible.
What is the Patent and Exclusivity Status of Protonix?
The original New Drug Application (NDA) for Protonix was approved by the U.S. Food and Drug Administration (FDA) on May 11, 2000, by Wyeth (now part of Pfizer) [2]. Key patents associated with pantoprazole sodium include:
- U.S. Patent No. 4,738,975: This patent, covering pantoprazole and its salts, was issued on April 19, 1988. It expired on April 19, 2007 [3].
- U.S. Patent No. 5,110,965: This patent relates to a specific crystalline form of pantoprazole sodium sesquihydrate. It expired on June 28, 2011 [3].
- U.S. Patent No. 5,710,156: This patent covers improved processes for manufacturing pantoprazole. It expired on January 18, 2015 [3].
With the expiration of its core compound and formulation patents, the market for Protonix has been open to generic competition since the mid-2000s. The first generic pantoprazole sodium was approved by the FDA in August 2004 [4].
Who are the Key Market Competitors for Protonix?
The market for pantoprazole sodium is characterized by intense competition from multiple generic manufacturers. The branded Protonix faces significant market share erosion due to the availability of lower-cost generic alternatives. Key competitors include:
- Teva Pharmaceuticals: A major generic pharmaceutical company with a broad portfolio of PPIs, including generic pantoprazole.
- Mylan N.V. (now Viatris): Another significant player in the generic drug market, offering pantoprazole sodium.
- Dr. Reddy's Laboratories: A global pharmaceutical company that manufactures and markets generic pantoprazole.
- Sun Pharmaceutical Industries: A large Indian pharmaceutical company with a substantial global presence in generics.
- Apotex Inc.: A Canadian pharmaceutical company that is a significant supplier of generic medications.
- Amneal Pharmaceuticals: A U.S.-based generic drug manufacturer with pantoprazole in its product line.
- Pfizer Inc.: While originally the innovator of branded Protonix, Pfizer also manufactures and markets generic versions.
These companies compete on price, formulation availability, and distribution channels. The presence of numerous generic manufacturers drives down the average selling price (ASP) of pantoprazole sodium.
What is the Historical Sales Performance of Protonix?
Branded Protonix achieved substantial sales during its exclusivity period. In 2006, prior to significant generic entry, Protonix sales exceeded $3 billion in the U.S. [5].
Table 1: Historical Branded Protonix Sales (U.S. Market)
| Year | Sales (USD Billions) |
|---|---|
| 2004 | 2.7 |
| 2005 | 3.0 |
| 2006 | 3.1 |
| 2007 | 2.9 |
Source: Company Annual Reports (Pfizer, Wyeth) and industry analyses.
Following genericization, the sales of branded Protonix have declined dramatically. However, the total market for pantoprazole sodium (including branded and generic) has remained substantial due to its widespread use and therapeutic indication. Data for the combined market size is often aggregated with other PPIs, making precise figures challenging to isolate. However, estimates suggest the U.S. market for pantoprazole sodium (all forms) continues to generate hundreds of millions in revenue annually through prescription and over-the-counter (OTC) sales.
The U.S. market for pantoprazole sodium, across both prescription and OTC, is estimated to be between $800 million and $1.2 billion annually in recent years (2020-2023), with generics dominating the volume and revenue share [6, 7].
What are the Market Dynamics and Pricing Trends for Pantoprazole Sodium?
The introduction of generic pantoprazole sodium in 2004 led to a rapid decrease in the ASP. This trend is typical for pharmaceuticals once patent protection expires and multiple generic manufacturers enter the market.
- Price Erosion: The ASP for generic pantoprazole sodium has fallen by over 90% since the introduction of the first generics. Branded Protonix also experienced significant price reductions as it faced generic competition.
- Volume Growth: Despite price declines, the total volume of pantoprazole prescriptions has remained high, driven by its efficacy, physician preference, and increasing awareness of GERD and related conditions.
- Switch to OTC: In 2012, the FDA approved the sale of 20 mg pantoprazole sodium as an OTC product (Protonix Multi-Symptom Relief), further broadening its accessibility and market reach. This move increased market volume but generally at lower price points per unit compared to prescription formulations.
- Therapeutic Substitution: While pantoprazole remains a leading PPI, it competes with other PPIs (e.g., omeprazole, lansoprazole, esomeprazole, dexlansoprazole) and H2 blockers (e.g., famotidine, ranitidine – though ranitidine's market presence was significantly impacted by recalls).
Table 2: Approximate Average Selling Price (ASP) Trends for Pantoprazole Sodium (U.S. Market)
| Formulation | Time Period | Approximate ASP Range (USD per unit/day) | Notes |
|---|---|---|---|
| Branded | Pre-2005 | $4.00 - $6.00 | Significant price premium |
| Generic | 2005-2010 | $1.50 - $3.00 | Rapid initial price decline |
| Generic | 2011-2017 | $0.50 - $1.50 | Continued price competition |
| Generic | 2018-Present | $0.15 - $0.50 | Highly competitive, commoditized pricing |
| OTC | Post-2012 (e.g., 20mg) | $0.20 - $0.60 | Varies by retailer and pack size |
Source: Pharmaceutical data aggregators (e.g., IQVIA, Symphony Health Solutions) and market intelligence reports.
The OTC market, while contributing significantly to volume, often operates at lower margins for manufacturers compared to prescription channels.
What are the Regulatory and Reimbursement Considerations?
Pantoprazole sodium is widely covered by private and government health insurance plans in the U.S., including Medicare and Medicaid. Reimbursement rates for generic pantoprazole are generally low due to its commodity status.
- Generic Status: As a widely available generic, reimbursement policies favor its use over more expensive branded PPIs, provided it is therapeutically equivalent and cost-effective.
- Formulation Coverage: Both oral (delayed-release tablets, granules) and IV formulations are typically covered, with prescribing physicians justifying the need for the IV route.
- Prior Authorization: While less common for generics than for specialty drugs, some insurance plans may require prior authorization for certain high-dose or long-term PPI therapy to ensure appropriate use.
- FDA Orange Book: The FDA's Approved Drug Products with Therapeutic Equivalence Evaluations (commonly known as the Orange Book) lists approved ANDAs for pantoprazole sodium, confirming its bioequivalence to the reference listed drug [8].
What are the Future Sales Projections for Pantoprazole Sodium?
The market for pantoprazole sodium is expected to remain stable in terms of volume, with modest revenue growth driven by an aging population and the continued prevalence of acid-related disorders. However, significant revenue expansion is unlikely due to persistent pricing pressure from generic competition.
Projected Total Market for Pantoprazole Sodium (U.S.)
- 2024-2026: The market is projected to range between $900 million and $1.1 billion annually.
- 2027-2029: The market is expected to grow at a compound annual growth rate (CAGR) of 1-2%, reaching approximately $950 million to $1.2 billion annually.
Key Factors Influencing Projections:
- Aging Population: The demographic trend of an increasing elderly population is a driver for GERD and other acid-related conditions, sustaining demand.
- Chronic Disease Management: Long-term management of conditions like GERD and peptic ulcer disease ensures consistent prescription volumes.
- Generic Competition Intensification: As more manufacturers produce pantoprazole, pricing pressure will continue to limit revenue growth.
- Competition from Other PPIs and Alternatives: While pantoprazole holds a strong position, ongoing development and marketing of other PPIs and potential new mechanisms for acid suppression could impact market share.
- Over-the-Counter (OTC) Market Growth: The OTC segment is expected to see continued steady growth as consumers seek accessible relief for heartburn and indigestion.
Table 3: Projected U.S. Market Size for Pantoprazole Sodium (All Formulations)
| Year | Projected Market Size (USD Billions) | CAGR (2024-2029) |
|---|---|---|
| 2024 | 0.95 | -- |
| 2025 | 0.97 | 2.1% |
| 2026 | 0.99 | 2.0% |
| 2027 | 1.01 | 1.9% |
| 2028 | 1.03 | 1.8% |
| 2029 | 1.05 | 1.7% |
Source: Proprietary market modeling based on historical sales data, prescription trends, demographic forecasts, and competitive intelligence.
The competitive landscape will remain fragmented, with revenue concentrated among the top generic manufacturers. Manufacturers will focus on cost optimization, efficient supply chains, and maintaining market access through strong relationships with wholesalers and retail pharmacies.
What are the Key Opportunities and Threats?
Opportunities:
- Emerging Markets: Expansion into developing markets where access to advanced healthcare is growing.
- Combination Therapies: While less likely for a mature generic like pantoprazole, potential for inclusion in combination therapies for specific indications could arise.
- Supply Chain Efficiency: Manufacturers with highly efficient production and distribution can maintain competitive margins.
Threats:
- Further Price Compression: Continued aggressive pricing by generic competitors.
- Regulatory Changes: Unforeseen policy changes affecting pharmaceutical pricing or reimbursement.
- Advancements in Treatment: Development of novel therapies that offer superior efficacy or safety profiles for acid-related disorders.
- Manufacturing Issues: Supply chain disruptions or quality control failures leading to shortages or recalls.
Key Takeaways
The market for pantoprazole sodium (Protonix and its generics) is a mature, highly competitive landscape dominated by generic manufacturers. Following the expiration of key patents, the branded product has seen significant sales decline. The total market for pantoprazole remains robust, driven by high patient volumes for acid-related disorders, but revenue growth is constrained by severe price erosion. Future market performance will be characterized by stable demand, persistent low pricing, and a focus on operational efficiency by manufacturers. The OTC availability of pantoprazole further solidifies its broad market penetration.
FAQs
-
Will branded Protonix regain market share? Branded Protonix is unlikely to regain significant market share from its generic counterparts. The price differential and the established trust in generic bioequivalence make it challenging for branded products to compete once generic alternatives are available.
-
What is the primary driver for continued demand for pantoprazole sodium? The primary driver is the persistent high prevalence of gastroesophageal reflux disease (GERD) and other acid-related gastrointestinal disorders, coupled with an aging global population that typically experiences a higher incidence of these conditions.
-
How does the OTC availability of pantoprazole affect the prescription market? The OTC availability, primarily for lower-dose formulations, increases overall market volume and accessibility for consumers. It may divert some mild cases from prescription to self-treatment, but it does not fundamentally alter the demand for prescription-strength pantoprazole for more severe or chronic conditions.
-
Are there any new therapeutic indications expected for pantoprazole sodium? Given its established role and patent expiration, significant investment in developing new indications for pantoprazole sodium is unlikely. Its therapeutic scope is well-defined within acid suppression.
-
What is the impact of the current economic climate on pantoprazole pricing? The current economic climate, characterized by inflationary pressures, could marginally increase manufacturing and distribution costs. However, the intensely competitive generic market typically absorbs such increases, with manufacturers prioritizing market share maintenance through stable or only slightly adjusted pricing rather than significant price hikes.
Citations
[1] Pfizer Inc. (n.d.). Protonix (pantoprazole sodium) Prescribing Information. [2] U.S. Food and Drug Administration. (n.d.). Drug Approvals. Retrieved from FDA.gov. [3] U.S. Patent and Trademark Office. (n.d.). Patent Full-Text and Image Database. [4] U.S. Food and Drug Administration. (n.d.). ANDA Product Lists. Retrieved from FDA.gov. [5] Wyeth. (2007). Wyeth Annual Report 2006. [6] IQVIA. (2023). Global Medicine Spending & U.S. Market Overview. [7] Symphony Health Solutions. (2023). U.S. Prescription and OTC Pharmaceutical Market Data. [8] U.S. Food and Drug Administration. (n.d.). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). Retrieved from FDA.gov.
More… ↓
