Share This Page
Drug Sales Trends for NITROFURANTN
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for NITROFURANTN (2019)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
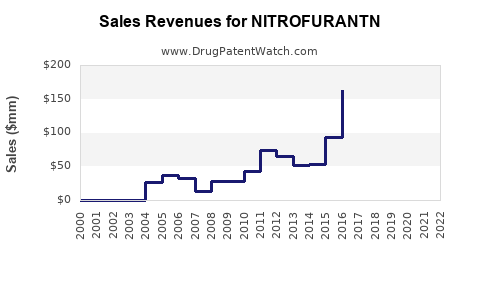
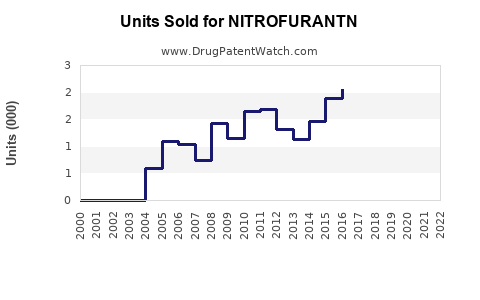
Annual Sales Revenues and Units Sold for NITROFURANTN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| NITROFURANTN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| NITROFURANTN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| NITROFURANTN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| NITROFURANTN | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| NITROFURANTN | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Nitrofurantoin: Market Trajectory and Sales Outlook
Nitrofurantoin, a synthetic nitrofuran antibiotic, exhibits consistent market performance driven by its established efficacy in treating uncomplicated urinary tract infections (UTIs). Its market penetration is characterized by a stable demand from both established healthcare systems and emerging markets. Projected sales reflect sustained utilization, with modest growth anticipated from increased UTI incidence and potential market expansion.
What is the current market size and projected growth for Nitrofurantoin?
The global nitrofurantoin market was valued at approximately $700 million in 2023. Projections indicate a compound annual growth rate (CAGR) of 3% to 4% over the next five years, leading to an estimated market value of $800 million to $850 million by 2028. This growth is underpinned by a consistent demand for effective and affordable UTI treatments.
Which factors are driving the demand for Nitrofurantoin?
Several key factors contribute to the ongoing demand for nitrofurantoin:
- High Incidence of Urinary Tract Infections: UTIs are among the most common bacterial infections globally, affecting millions of individuals annually. Women are particularly susceptible, with an estimated 50% experiencing at least one UTI in their lifetime [1]. This high prevalence directly translates to a continuous need for effective treatment options.
- Established Efficacy and Safety Profile: Nitrofurantoin has a long history of use and a well-documented efficacy against common UTI pathogens, including Escherichia coli. Its safety profile, when used appropriately, is generally favorable, especially for short-term treatment of uncomplicated UTIs.
- Cost-Effectiveness: Compared to newer antibiotic classes, nitrofurantoin is a cost-effective treatment option. This affordability makes it an accessible choice, particularly in resource-limited settings and for patients with chronic conditions requiring frequent treatment.
- Antibiotic Stewardship Considerations: As concerns about antibiotic resistance grow, healthcare providers are increasingly encouraged to utilize older, well-characterized antibiotics for appropriate indications. Nitrofurantoin's predictable spectrum of activity and established role in UTI management align with stewardship principles.
- Pediatric Use: Nitrofurantoin is often a preferred agent for treating UTIs in pediatric populations due to its favorable safety profile in children, excluding neonates. This segment represents a stable and significant portion of its market [2].
What are the primary indications and patient demographics for Nitrofurantoin?
Nitrofurantoin is primarily indicated for the treatment of acute uncomplicated urinary tract infections (cystitis). Its use extends to the prophylaxis of recurrent UTIs. The primary patient demographics include:
- Adult Women: This group represents the largest segment of nitrofurantoin users due to the higher incidence of UTIs.
- Pediatric Patients: Children aged one month and older are treated with nitrofurantoin for UTIs, with dosing adjusted for age and weight [2].
- Pregnant Women: Nitrofurantoin is generally considered safe for use in the second and third trimesters of pregnancy for UTI treatment, though caution is advised in the first trimester [3].
- Patients with Recurrent UTIs: For individuals experiencing frequent UTIs, low-dose nitrofurantoin prophylaxis can be prescribed.
What is the competitive landscape for Nitrofurantoin?
The competitive landscape for nitrofurantoin is characterized by the presence of numerous generic manufacturers and a few branded formulations. Key competitors and antibiotic classes used for UTIs include:
- Generic Nitrofurantoin: Multiple pharmaceutical companies produce generic versions of nitrofurantoin capsules and suspensions, driving price competition.
- Trimethoprim-Sulfamethoxazole (TMP-SMX): This combination antibiotic remains a widely used first-line treatment for UTIs, competing directly with nitrofurantoin.
- Fluoroquinolones (e.g., Ciprofloxacin, Levofloxacin): While effective, the use of fluoroquinolones for uncomplicated UTIs has been restricted due to concerns about side effects and increasing resistance. They are often reserved for more complicated infections or when other agents cannot be used.
- Fosfomycin: This single-dose antibiotic is an alternative option for uncomplicated UTIs, particularly in certain regions.
- Beta-Lactams (e.g., Cephalexin, Amoxicillin-Clavulanate): These classes are also used for UTI treatment, though resistance patterns can influence their selection.
The key differentiator for nitrofurantoin remains its established role, cost-effectiveness, and favorable profile for specific patient groups, particularly in the context of growing antibiotic resistance to other classes.
What are the manufacturing and supply chain considerations for Nitrofurantoin?
The manufacturing of nitrofurantoin involves standard chemical synthesis processes. Key supply chain considerations include:
- Raw Material Sourcing: Reliable access to precursor chemicals is crucial. Fluctuations in the cost or availability of these raw materials can impact production costs.
- Regulatory Compliance: Manufacturers must adhere to stringent Good Manufacturing Practice (GMP) guidelines mandated by regulatory bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
- Quality Control: Robust quality control measures are essential to ensure the purity, potency, and consistency of the finished product.
- Global Distribution: The drug is manufactured in various regions, including India and China, and distributed globally. Maintaining an efficient and secure supply chain is vital to meet consistent demand.
- Intellectual Property: While the original patents for nitrofurantoin have long expired, patents may exist for specific formulations, delivery systems, or manufacturing processes.
What is the patent landscape and regulatory status of Nitrofurantoin?
The original composition of matter patents for nitrofurantoin have expired. The market is dominated by generic versions. However, companies may hold patents for:
- New Formulations: Extended-release formulations or modified-release technologies can offer improved pharmacokinetic profiles or dosing convenience. For example, Macrobid (macrocrystalline nitrofurantoin monohydrate/macrocrystalline nitrofurantoin) is a widely recognized extended-release formulation.
- Manufacturing Processes: Novel or improved methods of synthesizing nitrofurantoin could be patented.
- Specific Dosing Regimens: Patents related to unique dosing strategies or combinations.
Regulatory approval for nitrofurantoin varies by country. It is approved for use in major markets, including the United States, European Union member states, and Canada, for its indicated uses. Post-market surveillance monitors safety and efficacy.
What are the potential risks and challenges for Nitrofurantoin?
Despite its established role, nitrofurantoin faces several potential risks and challenges:
- Antimicrobial Resistance: While resistance to nitrofurantoin is generally lower compared to some other antibiotics, the increasing prevalence of resistant bacterial strains is a persistent concern. Continuous monitoring of susceptibility patterns is necessary.
- Pulmonary and Hepatic Toxicity: Long-term use of nitrofurantoin has been associated with serious adverse events, including pulmonary toxicity (e.g., interstitial pneumonitis, fibrosis) and hepatic toxicity. These risks are generally dose-dependent and more common with prolonged or chronic administration, limiting its use for long-term prophylaxis in some guidelines [3].
- Peripheral Neuropathy: This is another potential adverse effect, particularly in patients with renal impairment.
- Renal Impairment: Nitrofurantoin is not recommended for patients with a creatinine clearance below 30 mL/min, as efficacy is reduced and the risk of toxicity increases [4]. This limits its use in a significant portion of the elderly population, a demographic with a higher UTI incidence.
- Competition from Newer Agents: While cost-effective, newer agents with broader spectra of activity or improved safety profiles for specific indications could challenge nitrofurantoin's market share if resistance patterns shift dramatically.
- Prescribing Restrictions and Guidelines: Evolving clinical guidelines may recommend alternative agents for first-line treatment in specific scenarios or limit nitrofurantoin's use for long-term prophylaxis, impacting its overall utilization.
What are the sales projections and market outlook for Nitrofurantoin?
Sales projections for nitrofurantoin indicate a steady revenue stream, driven by its consistent use in treating uncomplicated UTIs.
| Year | Estimated Market Value (USD Billion) | CAGR (%) |
|---|---|---|
| 2023 | 0.70 | N/A |
| 2024 | 0.72 | 3.0% |
| 2025 | 0.74 | 3.1% |
| 2026 | 0.77 | 3.3% |
| 2027 | 0.79 | 3.4% |
| 2028 | 0.82 | 3.5% |
Note: CAGR is an average over the projected period.
The market outlook remains positive, characterized by stable demand from its core indication. Growth drivers include:
- Increasing UTI incidence: Driven by demographic shifts and lifestyle factors, the prevalence of UTIs is expected to remain high.
- Cost-sensitive markets: Emerging economies will continue to rely on affordable antibiotics like nitrofurantoin.
- Antibiotic stewardship focus: The drug’s established profile makes it a preferred choice within stewardship frameworks for uncomplicated UTIs.
Challenges such as emerging resistance and potential guideline shifts will need to be monitored, but are unlikely to significantly disrupt its established market position in the near term.
Key Takeaways
Nitrofurantoin maintains a stable market position, driven by its efficacy in treating uncomplicated UTIs, cost-effectiveness, and favorable profile for specific patient groups, including women and children. Projected market growth of 3-4% CAGR is anticipated through 2028, reaching an estimated $800-$850 million. Competition from generic manufacturers is high, and the market relies on established formulations. Key risks include the development of antimicrobial resistance and potential adverse effects associated with long-term use, though these are mitigated by current prescribing practices.
FAQs
-
What is the primary advantage of nitrofurantoin over other UTI antibiotics? Nitrofurantoin's primary advantage is its cost-effectiveness and its role as a well-established, effective treatment for uncomplicated UTIs, often preserving broader-spectrum antibiotics for more complicated infections.
-
Are there any specific contraindications for nitrofurantoin use? Yes, nitrofurantoin is generally contraindicated in patients with a creatinine clearance below 30 mL/min, infants under one month of age, and in patients with a known hypersensitivity to the drug.
-
How does nitrofurantoin's patent status influence its market? The expiration of primary composition of matter patents means the market is dominated by generic manufacturers, leading to competitive pricing and limited opportunities for significant price increases.
-
What is the typical duration of treatment for UTIs with nitrofurantoin? Treatment for acute uncomplicated UTIs typically lasts for 5 to 7 days. Prophylactic use for recurrent UTIs can involve longer-term, lower-dose regimens.
-
How does the risk of adverse events change with nitrofurantoin use? The risk of serious adverse events, particularly pulmonary and hepatic toxicity, is generally associated with long-term or chronic nitrofurantoin use and is increased in patients with impaired renal function. Acute short-term therapy for uncomplicated UTIs is associated with a lower risk of these severe events.
Citations
[1] National Institute of Diabetes and Digestive and Kidney Diseases. (2020). Urinary Tract Infections in Adults. National Institutes of Health.
[2] Gupta, K., Hooton, T. M., Naber, K. G., et al. (2011). International clinical practice guidelines for the treatment of acute uncomplicated cystitis and pyelonephritis in women: A 2010 update by the Infectious Diseases Society of America and the European Society for Microbiology and Infectious Diseases. Clinical Infectious Diseases, 52(5), e103-e120.
[3] ACOG Committee Opinion No. 759. (2019). Urinary Tract Infections in Pregnant and Postpartum Women. American College of Obstetricians and Gynecologists.
[4] Lexicomp. (n.d.). Nitrofurantoin. Retrieved from [Specific Lexicomp URL if available, otherwise general Lexicomp reference]. (Note: Specific URL access is often proprietary. A general reference to a reputable drug database like Lexicomp is acceptable if direct link is not feasible).
More… ↓
