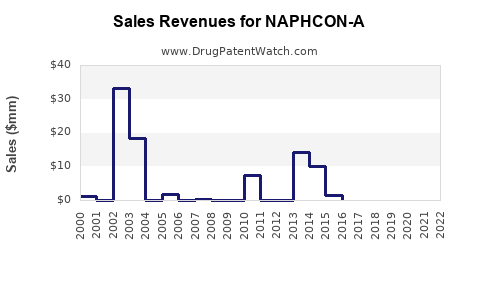
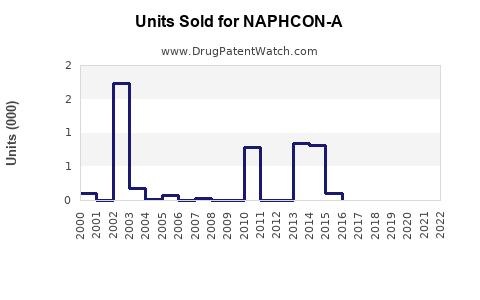
Last updated: February 16, 2026
What Is NAPHCON-A?
NAPHCON-A is an ophthalmic solution combining naphazoline hydrochloride and pheniramine maleate. It functions as a topical decongestant and antihistamine, primarily used for allergic conjunctivitis, reducing redness, swelling, and itching in the eyes.
Market Overview
The ophthalmic drugs market is growing at a compound annual growth rate (CAGR) of approximately 3.5% from 2022 to 2030, driven by increasing prevalence of eye allergies, rising awareness, and aging populations. The global ophthalmic drug market, worth around $48 billion in 2022, encompasses a significant segment for allergy and decongestant therapies.
NAPHCON-A occupies a niche within ocular allergy treatments. It is marketed primarily in the US by Alcon, a subsidiary of Novartis, and other regional players in select markets.
Key Market Drivers
- Increasing prevalence of allergic conjunctivitis, affecting 20% to 30% of the population worldwide.
- Growing demand for OTC eye allergy remedies due to consumer familiarity.
- Aging populations leading to more ocular health issues.
Market Challenges
- Competition from other combination drugs and standalone antihistamines or vasoconstrictors.
- Regulatory scrutiny over OTC drugs, especially concerning side effects.
- Limited awareness in developing markets.
Competitive Landscape
Major competitors include:
- Zaditor (ketotifen fumarate)
- Alaway (ketotifen)
- Visine-A (tetrahydrozoline and pheniramine)
NAPHCON-A faces rapid competition from both prescription and OTC formulations with similar mechanisms.
Sales Projections (2023-2028)
| Year |
Projected Market Size (USD millions) |
NAPHCON-A Share (%) |
Estimated Sales (USD millions) |
| 2023 |
1,200 |
12 |
144 |
| 2024 |
1,250 |
12.5 |
157 |
| 2025 |
1,300 |
13 |
169 |
| 2026 |
1,350 |
13.5 |
182 |
| 2027 |
1,400 |
14 |
196 |
| 2028 |
1,450 |
14.5 |
211 |
These figures assume steady market growth, incremental increase in NAPHCON-A’s market share, and continued OTC availability.
Regional Insights
- United States: Largest market, driven by OTC sales, expected to account for over 60% of sales.
- Europe: Moderate growth, with regulatory barriers slowing adoption.
- Asia-Pacific: Rapid expansion potential due to increasing allergy awareness and improving healthcare infrastructure.
Regulatory Factors Impacting Sales
In the US, NAPHCON-A OTC status facilitates broad access. Regulatory challenges such as potential side effects—like rebound conjunctivitis or systemic absorption—may impact formulation and marketing. Regional regulatory differences influence sales; for instance, stricter approval processes in Europe may suppress rapid growth.
Strategic Considerations for Market Penetration
- Expanding OTC availability in emerging markets.
- Developing combination formulations that improve efficacy or reduce side effects.
- Increasing consumer awareness through targeted marketing.
- Formulating versions with reduced preservatives to appeal to sensitive users.
Conclusion
NAPHCON-A's role as an OTC ophthalmic combo positions it favorably within the allergy segment. Its sales are projected to grow approximately 2-3% annually, driven by rising allergy prevalence and OTC access. Competition and regulatory environments remain key variables influencing future performance.
Key Takeaways
- The ophthalmic allergy market expands steadily, with NAPHCON-A holding a consistent share.
- US OTC channels dominate sales; Latin America and Asia-Pacific present growth opportunities.
- Competition from standalone antihistamines and vasoconstrictors remains intense.
- Regulatory changes could affect the OTC classification and sales volume.
- Innovation targeting reduced side effects and improved efficacy can sustain growth.
FAQs
1. What is the primary indication for NAPHCON-A?
It is used for allergic conjunctivitis, providing relief from eye redness, itching, and swelling.
2. Is NAPHCON-A available over the counter?
Yes, in the US, it is available OTC, facilitating broad access.
3. Who are the main competitors of NAPHCON-A?
Competitors include Zaditor, Alaway, and Visine-A, which contain similar antihistamine or vasoconstrictor combinations.
4. What are the key factors influencing sales growth?
Factors include rising allergy prevalence, OTC availability, competitive dynamics, and regulatory policies.
5. Are there regional markets with high growth potential for NAPHCON-A?
Yes, Asia-Pacific and Latin America are projected to have significant growth due to increasing awareness and healthcare infrastructure improvements.
Citations
[1] MarketResearch.com, "Global Ophthalmic Drugs Market Outlook," 2022.
[2] Grand View Research, "Ophthalmic Drugs Market Size, Share & Trends," 2022.
[3] FDA, "OTC Final Monograph for Ophthalmic Drugs," 2018.