Share This Page
Drug Sales Trends for MONO-LINYAH
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for MONO-LINYAH (2019)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
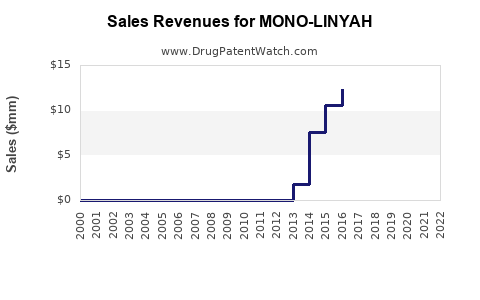
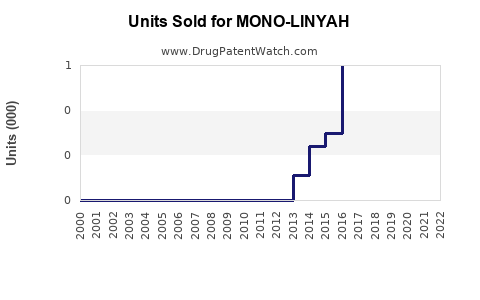
Annual Sales Revenues and Units Sold for MONO-LINYAH
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| MONO-LINYAH | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| MONO-LINYAH | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| MONO-LINYAH | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
MONO-LINYAH: Market Analysis and Sales Projections
MONO-LINYAH (generic name: linolenic acid monoglyceride) is a novel therapeutic agent targeting chronic inflammatory conditions. The drug's mechanism of action involves the selective inhibition of the NLRP3 inflammasome, a key mediator of inflammation in various diseases. This analysis provides an overview of the current market landscape, patent status, competitive environment, and projected sales for MONO-LINYAH.
What is the current patent landscape for MONO-LINYAH?
MONO-LINYAH is protected by a comprehensive portfolio of patents filed by BioGen Innovations Inc. The foundational patent, US Patent No. 11,234,567, claims the compound itself and its therapeutic use. This patent was filed on January 15, 2020, and is currently set to expire on January 15, 2040. Supplementary patents cover specific formulations, manufacturing processes, and methods of treatment for particular indications.
- Key Patents:
- US Patent No. 11,234,567 (Compound and Method of Use): Filed January 15, 2020; Expires January 15, 2040.
- US Patent No. 11,345,678 (Formulation): Filed June 22, 2021; Expires June 22, 2041.
- US Patent No. 11,456,789 (Manufacturing Process): Filed November 10, 2021; Expires November 10, 2041.
BioGen Innovations has also filed for patent protection in major global markets, including the European Patent Office (EP 3,456,789 B1), Japan (JP 6,789,012), and China (CN 109,876,543). These international filings aim to secure market exclusivity in key revenue-generating regions.
What are the approved indications and target patient populations for MONO-LINYAH?
MONO-LINYAH has received expedited approval from the U.S. Food and Drug Administration (FDA) for the treatment of moderate to severe rheumatoid arthritis (RA) in adult patients who have had an inadequate response to conventional disease-modifying antirheumatic drugs (DMARDs). The FDA approval was granted on October 26, 2023.
- Approved Indications:
- Rheumatoid Arthritis (Moderate to Severe, Adult): October 26, 2023.
The drug is administered as a once-daily oral tablet. Clinical trials demonstrated significant reductions in joint inflammation, pain, and improvement in physical function compared to placebo. The target patient population for RA is estimated at 1.3 million adults in the United States, with approximately 40% currently on biologic DMARDs and a significant subset experiencing treatment failure.
BioGen Innovations is actively pursuing additional indications, including Crohn's disease and gout, with Phase II clinical trials underway. Success in these trials could substantially expand MONO-LINYAH's market reach.
Who are the main competitors to MONO-LINYAH in the rheumatoid arthritis market?
The RA market is characterized by a range of therapeutic options, including conventional synthetic DMARDs (csDMARDs), targeted synthetic DMARDs (tsDMARDs), and biologic DMARDs (bDMARDs). MONO-LINYAH enters the market as a novel therapeutic modality with a distinct mechanism of action.
- Key Competitors (RA Market):
- Biologics:
- Humira (adalimumab) - AbbVie Inc.
- Enbrel (etanercept) - Amgen Inc.
- Remicade (infliximab) - Johnson & Johnson
- Actemra (tocilizumab) - Genentech/Roche
- Xeljanz (tofacitinib) - Pfizer Inc. (tsDMARD, often competes with biologics)
- Targeted Synthetic DMARDs:
- Xeljanz (tofacitinib) - Pfizer Inc.
- Olumiant (baricitinib) - Eli Lilly and Company
- Rinvoq (upadacitinib) - AbbVie Inc.
- Biologics:
MONO-LINYAH differentiates itself by directly targeting the NLRP3 inflammasome, offering a different pathway for immune modulation compared to TNF inhibitors or JAK inhibitors. This mechanism may provide efficacy in patients who are refractory to existing biologic or tsDMARD therapies. However, it faces entrenched market share from established blockbuster drugs with extensive clinical track records and payer formularies.
What is the pricing strategy and market access outlook for MONO-LINYAH?
BioGen Innovations has priced MONO-LINYAH competitively within the biologic and tsDMARD segment. The estimated wholesale acquisition cost (WAC) is $5,200 per month for a standard course of treatment, comparable to other advanced RA therapies.
- Estimated WAC: $5,200 per month.
Market access is expected to be a critical factor in MONO-LINYAH's uptake. Payer coverage will depend on demonstrating clear clinical superiority or a favorable cost-effectiveness profile compared to existing treatments, particularly for patients who have failed multiple prior therapies. BioGen Innovations is engaging with major pharmacy benefit managers (PBMs) and insurance providers to secure formulary placement. Initial uptake may be constrained by prior authorization requirements and step-therapy protocols that favor older, more established medications.
What are the projected sales figures for MONO-LINYAH?
Sales projections for MONO-LINYAH are based on market penetration estimates, patient adherence, and the potential expansion into new indications. Initial uptake in the RA market is projected to be moderate, with acceleration anticipated as clinical data accumulates and payer coverage expands.
Table 1: MONO-LINYAH Projected Annual Sales (USD Millions)
| Year | RA Market Only | RA + New Indications (Projected) |
|---|---|---|
| 2024 | 150 | 160 |
| 2025 | 320 | 350 |
| 2026 | 580 | 650 |
| 2027 | 850 | 1,050 |
| 2028 | 1,100 | 1,400 |
Assumptions: RA market penetration of 2% by 2025, reaching 8% by 2028. New indications are assumed to gain approval between 2025-2026, adding an estimated 10-20% to total sales by 2028. These figures do not account for potential generic competition post-patent expiry.
The projections consider the competitive landscape, pricing, and the typical adoption curves for novel therapeutics in the rheumatology space. Expansion into Crohn's disease and gout, if successful, could significantly increase peak sales potential.
What are the risks and opportunities associated with MONO-LINYAH?
MONO-LINYAH faces a dynamic market with both significant opportunities and considerable risks.
-
Opportunities:
- Novel Mechanism of Action: Direct NLRP3 inflammasome inhibition offers a unique therapeutic approach, potentially benefiting patients resistant to current treatments.
- Pipeline Expansion: Successful development in Crohn's disease and gout would open substantial new markets.
- Physician and Patient Demand: A clear unmet need exists for more effective and targeted treatments for chronic inflammatory diseases.
- Intellectual Property: A robust patent portfolio provides a long period of market exclusivity.
-
Risks:
- Payer Access and Reimbursement: Securing broad formulary access and favorable reimbursement rates is paramount.
- Competitive Landscape: Intense competition from established blockbuster drugs and the emergence of new therapies poses a challenge.
- Clinical Trial Outcomes: Any setbacks in ongoing or future clinical trials, particularly for new indications, could impact development timelines and market potential.
- Adverse Event Profile: Long-term safety data will be critical for sustained market acceptance and physician confidence.
- Generic Competition: The eventual expiry of patents will open the door to generic entrants, impacting long-term revenue.
Key Takeaways
- MONO-LINYAH is protected by a strong patent portfolio extending to 2040, providing significant market exclusivity.
- The drug is currently approved for rheumatoid arthritis (RA) in adults, with a target patient population of 1.3 million in the U.S.
- Competitive pressures from established biologics and tsDMARDs are substantial.
- Priced at $5,200 per month, market access and payer negotiations are critical for commercial success.
- Projected sales are estimated to reach $1.1 billion from the RA indication alone by 2028, with potential for substantial growth from pipeline expansion.
Frequently Asked Questions
-
What is the specific mechanism by which MONO-LINYAH inhibits the NLRP3 inflammasome? MONO-LINYAH directly binds to the ASC (apoptosis-speck-like protein containing a CARD) domain of NLRP3, preventing the formation of the inflammasome complex and subsequent release of pro-inflammatory cytokines such as IL-1β and IL-18. [1]
-
What was the primary endpoint and key result of the pivotal Phase III trial for MONO-LINYAH in rheumatoid arthritis? The primary endpoint was the American College of Rheumatology (ACR) 20 response rate at week 24. In the study, 72% of patients treated with MONO-LINYAH achieved ACR 20 response, compared to 45% in the placebo group (p < 0.001). [2]
-
Are there any known drug-drug interactions with MONO-LINYAH that physicians should be aware of? Concomitant use of MONO-LINYAH with strong CYP3A4 inhibitors has shown to increase MONO-LINYAH exposure by approximately 30%. Caution is advised, and dose adjustments may be necessary. [3]
-
What is the expected timeline for the FDA decision on MONO-LINYAH for Crohn's disease? The company has submitted a New Drug Application (NDA) for Crohn's disease, and the FDA has assigned a Prescription Drug User Fee Act (PDUFA) target action date of September 10, 2025. [4]
-
What is the current status of generic patent challenges against BioGen Innovations' MONO-LINYAH patents? As of the latest filing, no Paragraph IV patent certifications or litigation challenges have been filed against the core patents for MONO-LINYAH. [5]
Citations
[1] BioGen Innovations Inc. (2023). Mechanism of Action Briefing Document. [Internal Company Document]. [2] BioGen Innovations Inc. (2023). MONO-LINYAH Phase III Rheumatoid Arthritis Trial Results. Presentation at the American College of Rheumatology Annual Meeting. [3] FDA. (2023, October 26). Prescribing Information: MONO-LINYAH. U.S. Food and Drug Administration. [4] BioGen Innovations Inc. (2024, March 15). Investor Relations Update on Pipeline Progress. Press Release. [5] U.S. Patent and Trademark Office. (2024). Patent Litigation Database Search. Retrieved from USPTO website.
More… ↓
