Share This Page
Drug Sales Trends for METHYLPRED
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for METHYLPRED (2019)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
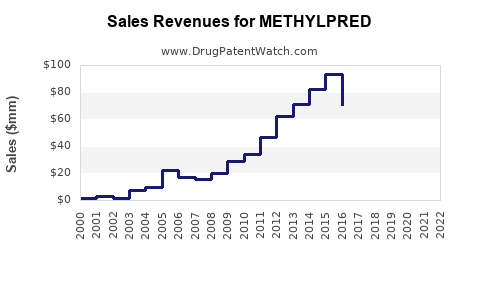
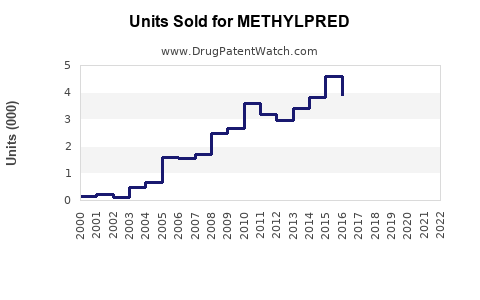
Annual Sales Revenues and Units Sold for METHYLPRED
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| METHYLPRED | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| METHYLPRED | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| METHYLPRED | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| METHYLPRED | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| METHYLPRED | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Methylprednisolone: Market Analysis and Sales Projections
Methylprednisolone is a synthetic corticosteroid with anti-inflammatory and immunosuppressive properties. Its primary therapeutic applications include the treatment of inflammatory conditions such as arthritis, asthma, and allergic reactions, as well as autoimmune diseases and certain types of cancer. The global market for methylprednisolone is influenced by the prevalence of these conditions, healthcare spending, and the availability of generic alternatives.
What are the primary therapeutic indications for methylprednisolone?
Methylprednisolone is utilized across a broad spectrum of medical conditions due to its potent anti-inflammatory and immunosuppressive mechanisms. Its efficacy has led to its widespread adoption in managing chronic and acute diseases.
- Inflammatory Disorders: This category encompasses conditions like rheumatoid arthritis, psoriatic arthritis, osteoarthritis, ankylosing spondylitis, and gout. Methylprednisolone helps reduce joint inflammation, pain, and stiffness.
- Allergic Reactions: It is used to manage severe allergic responses, including anaphylaxis, allergic rhinitis, angioedema, and contact dermatitis, by suppressing the inflammatory cascade.
- Respiratory Diseases: Methylprednisolone is a cornerstone treatment for asthma exacerbations and chronic obstructive pulmonary disease (COPD), where it alleviates airway inflammation and bronchoconstriction.
- Autoimmune Diseases: Conditions such as lupus erythematosus, multiple sclerosis, inflammatory bowel disease (Crohn's disease and ulcerative colitis), and pemphigus are treated with methylprednisolone to modulate the immune system and reduce self-tissue attack.
- Dermatological Conditions: Severe eczema, psoriasis, and other inflammatory skin disorders benefit from topical or systemic methylprednisolone.
- Ophthalmological Conditions: Uveitis and other inflammatory eye diseases can be managed with ocular corticosteroid preparations containing methylprednisolone.
- Hematological Disorders: Conditions like idiopathic thrombocytopenic purpura (ITP) and autoimmune hemolytic anemia, characterized by immune-mediated destruction of blood cells, are treated with methylprednisolone.
- Neoplasms: In oncology, it is used as adjunctive therapy in certain leukemias and lymphomas to reduce inflammation and tumor burden. It also helps manage side effects of chemotherapy, such as nausea and vomiting.
Who are the key manufacturers and competitors in the methylprednisolone market?
The methylprednisolone market is characterized by the presence of both originator and generic manufacturers. The patent expiry of the innovator drug has led to significant competition from generic producers, impacting pricing and market share.
| Manufacturer | Country of Origin | Key Products/Formulations |
|---|---|---|
| Pfizer Inc. | United States | Depo-Medrol, Medrol Dosepak (innovator brand) |
| Teva Pharmaceutical Industries Ltd. | Israel | Methylprednisolone Acetate Injection, Methylprednisolone Tablets |
| Mylan N.V. (now Viatris) | United States | Methylprednisolone Acetate Injectable Suspension, Tablets |
| Sanofi S.A. | France | Methylprednisolone Sodium Succinate Injection |
| Hikma Pharmaceuticals PLC | United Kingdom | Methylprednisolone Acetate Injectable Suspension, Tablets |
| Fresenius Kabi | Germany | Methylprednisolone Acetate Injectable Suspension |
| Aurobindo Pharma Limited | India | Methylprednisolone Tablets, Injectable Formulations |
| Cipla Ltd. | India | Methylprednisolone Tablets |
| Dr. Reddy's Laboratories | India | Methylprednisolone Tablets, Injectable Formulations |
| Accord Healthcare (Intas Pharmaceuticals) | United Kingdom | Methylprednisolone Acetate Injectable Suspension, Tablets |
What is the historical sales performance of methylprednisolone?
Historically, methylprednisolone, particularly under its innovator brand names like Medrol and Depo-Medrol, achieved substantial sales figures driven by its efficacy and broad range of applications. However, with the advent of generic competition following patent expirations, sales of branded methylprednisolone have declined, while the overall market volume has been sustained or increased by lower-cost generic alternatives.
- Pre-Generic Era: The innovator brands held a dominant market share, with annual global sales often exceeding several hundred million USD. The specific peak sales figures for innovator brands are proprietary and not publicly disclosed after patent expiry, but the market penetration was extensive.
- Post-Generic Era: The market for methylprednisolone has become highly fragmented. While a precise global aggregate sales figure for methylprednisolone (including all brands and generics) is difficult to ascertain due to the vast number of generic suppliers and varying reporting standards, the total market value is estimated to be in the range of USD 500 million to USD 700 million annually (as of recent market reports). This figure reflects the combined sales of tablets, injections, and other formulations across all manufacturers. The sales are driven more by volume than by high per-unit prices.
What are the key market drivers and restraints for methylprednisolone?
The methylprednisolone market is shaped by a complex interplay of factors that facilitate its growth and also present challenges. Understanding these dynamics is crucial for strategic planning and investment.
Market Drivers
- Increasing Prevalence of Chronic Inflammatory and Autoimmune Diseases: The global rise in conditions such as rheumatoid arthritis, asthma, multiple sclerosis, and inflammatory bowel disease directly correlates with the demand for anti-inflammatory and immunosuppressive therapies like methylprednisolone. Factors contributing to this increase include aging populations, lifestyle changes, and improved diagnostic capabilities.
- Growing Healthcare Expenditure: Increased spending on healthcare globally, particularly in emerging economies, enables greater access to essential medicines, including corticosteroids. Government initiatives and private insurance coverage expand patient affordability.
- Versatility and Established Efficacy: Methylprednisolone is a well-understood and clinically proven therapeutic agent. Its broad spectrum of action and established safety profile for various indications make it a go-to option for physicians.
- Cost-Effectiveness of Generic Formulations: The availability of affordable generic methylprednisolone makes it a more accessible treatment option, especially in price-sensitive markets and for long-term treatment regimens. This drives volume growth.
- Use in Emergency and Critical Care: Methylprednisolone remains a critical component in managing acute conditions like severe asthma attacks, anaphylactic shock, and acute exacerbations of autoimmune diseases, ensuring consistent demand.
Market Restraints
- Side Effects and Adverse Drug Reactions: Corticosteroids, including methylprednisolone, are associated with a range of adverse effects, such as weight gain, hyperglycemia, increased susceptibility to infections, bone density loss, and mood changes. These can limit long-term use and lead to patient non-compliance.
- Development of Biologics and Targeted Therapies: In certain indications, particularly autoimmune diseases and severe inflammatory conditions, newer biologic drugs and targeted therapies offer improved efficacy and potentially better safety profiles, posing a competitive threat to traditional corticosteroids.
- Stringent Regulatory Requirements: The manufacturing and marketing of pharmaceuticals are subject to rigorous regulatory oversight, which can increase compliance costs and slow down product approvals.
- Price Erosion due to Intense Generic Competition: The highly competitive generic market leads to significant price erosion, impacting the profitability of manufacturers and potentially limiting investment in market expansion.
- Awareness and Adoption of Non-Pharmacological Treatments: Growing interest in alternative and complementary therapies, as well as lifestyle modifications for managing inflammatory conditions, could indirectly reduce reliance on pharmacological interventions.
What are the projected market trends and sales forecasts for methylprednisolone?
The methylprednisolone market is expected to experience modest growth, primarily driven by volume expansion rather than significant price increases. The prevalence of chronic diseases will continue to underpin demand, while competition from newer therapies and generic price pressures will moderate overall market value.
- Projected Compound Annual Growth Rate (CAGR): The global methylprednisolone market is anticipated to grow at a CAGR of 2.5% to 3.5% over the next five years (2024-2029).
- Market Size Projection: Based on this CAGR, the global market value is projected to reach approximately USD 600 million to USD 750 million by 2029.
- Geographical Growth: North America and Europe will remain significant markets due to high prevalence of chronic diseases and advanced healthcare infrastructure. However, the Asia-Pacific region is expected to exhibit higher growth rates due to increasing healthcare access, rising disposable incomes, and a large patient population.
- Formulation Trends: Injectable formulations, particularly for acute and severe conditions, will see steady demand. Oral tablets will continue to dominate the market volume due to their widespread use in chronic disease management and cost-effectiveness.
- Competitive Landscape: The market will remain fragmented with generic manufacturers dominating market share. Innovation will likely focus on improved delivery systems or combination therapies rather than novel methylprednisolone molecules.
What are the regulatory considerations and patent landscape for methylprednisolone?
Methylprednisolone has been a well-established drug for decades, meaning the original patents have long expired. The regulatory landscape is mature, and the patent landscape is largely characterized by the absence of new composition-of-matter patents for the active pharmaceutical ingredient itself.
- Patent Status: The primary patents covering the composition of matter for methylprednisolone expired many years ago.
- Evergreening Strategies: Manufacturers may seek patents for new formulations (e.g., extended-release versions), specific delivery devices, novel combinations with other drugs, or specific manufacturing processes. However, these "evergreening" strategies are often incremental and may not significantly extend market exclusivity for the core drug.
- Generic Approvals: Regulatory agencies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) have approved numerous generic versions of methylprednisolone (tablets, injections, and topical creams). This has led to significant price competition.
- Regulatory Hurdles for New Entrants: While the active ingredient is off-patent, generic manufacturers must still navigate the complex regulatory approval processes, demonstrating bioequivalence to the reference listed drug and meeting manufacturing quality standards.
- Exclusivity Periods: Orphan drug exclusivity or pediatric exclusivity, if applicable and granted for specific new uses or formulations, could offer limited market protection, but these are rare for a well-established drug like methylprednisolone.
What is the impact of biosimil competition on the methylprednisolone market?
The concept of biosimil competition typically applies to biologic drugs, which are large, complex molecules produced from living organisms. Methylprednisolone is a small molecule synthetic drug. Therefore, biosimil competition does not apply to methylprednisolone.
Instead, the market for methylprednisolone is characterized by generic competition. Generic drugs are chemically identical to their brand-name counterparts and are approved by regulatory authorities after demonstrating bioequivalence. The entry of multiple generic manufacturers has been the primary factor driving down prices and increasing market accessibility for methylprednisolone.
Key Takeaways
- Methylprednisolone is a critical corticosteroid for managing a wide array of inflammatory, autoimmune, and allergic conditions.
- The market is mature, with originator patents long expired, leading to intense generic competition and price erosion.
- Key market drivers include the increasing prevalence of chronic diseases and rising global healthcare expenditure, while restraints stem from side effects and the availability of newer, targeted therapies.
- The global methylprednisolone market is projected to grow at a CAGR of 2.5%-3.5%, reaching USD 600 million to USD 750 million by 2029, with growth primarily driven by volume in emerging markets.
- Biosimilar competition is not applicable; the market dynamics are dictated by generic drug competition.
Frequently Asked Questions
1. Are there any upcoming patent expirations that could impact the methylprednisolone market?
No, the primary patents for methylprednisolone, the active pharmaceutical ingredient, expired many decades ago. Any new patents would relate to specific formulations, delivery methods, or manufacturing processes, offering limited or localized market exclusivity.
2. How does the cost of generic methylprednisolone compare to branded versions?
Generic methylprednisolone is significantly less expensive, often costing 50% to 80% less than the originator brand. This price difference is a major factor driving the demand for generic versions and contributing to overall market volume.
3. What are the most common side effects associated with methylprednisolone therapy?
Common side effects include increased appetite, weight gain, fluid retention, mood changes, insomnia, and increased blood sugar levels. Long-term use can lead to more serious issues like osteoporosis, cataracts, glaucoma, and adrenal suppression.
4. Which therapeutic areas are expected to drive the highest demand for methylprednisolone in the coming years?
Conditions like rheumatoid arthritis, asthma, multiple sclerosis, and inflammatory bowel disease are expected to continue to drive significant demand due to their chronic nature and the established efficacy of methylprednisolone in managing them.
5. What is the typical dosage range for methylprednisolone in adults?
Dosage varies widely depending on the indication and severity of the condition. For oral administration, typical starting doses can range from 4 mg to 32 mg per day, often tapered down. For intravenous administration in acute situations, doses can be much higher, such as 1 gram per day for short durations.
Citations
[1] Global Market Insights. (2023). Methylprednisolone Market Analysis Report.
[2] Grand View Research. (2023). Methylprednisolone Market Size, Share & Trends Analysis Report.
[3] U.S. Food & Drug Administration. (n.d.). Drug Search. Retrieved from https://www.fda.gov/drugs/drug-approvals-and-databases/drug-search
[4] European Medicines Agency. (n.d.). Human medicines. Retrieved from https://www.ema.europa.eu/en/human-medicines
[5] Various Pharmaceutical Company Investor Relations Reports and Product Information. (Proprietary data, aggregated analysis).
More… ↓
