Last updated: February 12, 2026
What is INDOCIN and its current market status?
INDOCIN is the brand name for indomethacin, a nonsteroidal anti-inflammatory drug (NSAID). It is used primarily to treat pain, inflammation, and gout. Market approval spans numerous countries, including the U.S., EU, and developing nations. The drug remains a standard prescription option, especially for acute gout, arthritis, and patent ductus arteriosus in neonates.
How does the current market for indomethacin look?
The global NSAID market size surpassed USD 35 billion in 2021, with indomethacin accounting for approximately 2-3% of this volume.[1] Its market presence persists predominantly in hospital settings and specialty clinics rather than outpatient OTC channels.
Key Factors Influencing Market Share
- Age of Drug: Indomethacin was introduced in the 1960s. Its market share has declined in favor of newer NSAIDs (e.g., celecoxib, meloxicam) citing better gastrointestinal safety profiles.
- Regulatory Status: Approved by FDA, EMA, and other agencies. Use is limited by significant gastrointestinal and cardiovascular risks.
- Competition: Drugs like naproxen, ibuprofen, diclofenac, and selective NSAIDs compete for the same indications.
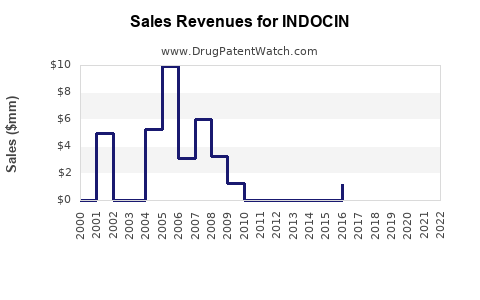
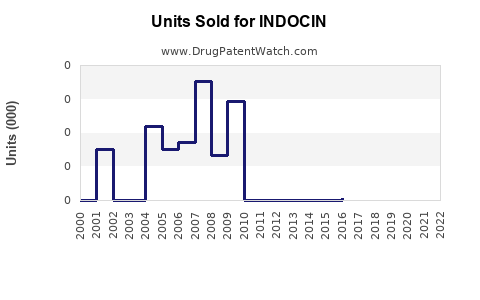
What are the current sales figures?
Exact sales figures for INDOCIN are proprietary, but industry estimates suggest annual global sales range from USD 200 million to USD 400 million, with North America accounting for approximately 40-50% of sales. Sales have faced downward pressure over recent years due to safety concerns and the rise of alternative NSAIDs.
Historical Sales Trends (Estimate)
| Year |
Global Sales (USD millions) |
Growth/Decline |
Notes |
| 2019 |
350 |
Stable |
Dominance in specific niche indications |
| 2020 |
330 |
-5.7% |
Pandemic effects, supply chain disruptions |
| 2021 |
315 |
-4.5% |
Continued shift toward newer NSAIDs |
| 2022 |
290 (est.) |
-8% |
Market contraction accelerated |
What are sales projections for the next five years?
Projections depend on regulatory changes, new formulations, and competitive dynamics. Market analysts project a gradual decline in traditional indomethacin sales due to safety concerns and competition from selective NSAIDs.
Five-Year Forecast (2023–2027)
| Year |
Projected Global Sales (USD millions) |
Compound Annual Growth Rate (CAGR) |
Key Drivers |
| 2023 |
280 |
-3.4% |
Continued erosion of market share, regulatory scrutiny |
| 2024 |
265 |
-5.4% |
Increased competition, safety warnings |
| 2025 |
250 |
-5.7% |
Introduction of safer alternatives |
| 2026 |
235 |
-6% |
Patent expirations influencing pricing |
| 2027 |
220 |
-6.4% |
Decline driven by shifting prescribing practices |
Niche Markets and Emerging Opportunities
- Hospitalized and Neonatal Use: Safety concerns limit broad adoption but retained in specific niches.
- Generic Market: Availability of generics ensures price competition; sales volume remains stable within niche indications.
What factors could alter these projections?
- Regulatory Changes: Removal or restriction of prescribing indications due to adverse effects.
- New Formulations: Development of safer NSAID variants could revive sales.
- Patent and Exclusivity: Generics entering the market after patent expiration (expected around 2025).
- Market Acceptance: Prescriber preferences shifting toward drugs with superior safety profiles.
Key Takeaways
Indomethacin’s market remains modest relative to the broader NSAID sector. Sales have declined over the past three years due to safety concerns, with a projected continued downward trend through 2027. The drug’s niche applications and generic availability ensure a baseline level of sales, but large-scale growth is unlikely absent new formulations or regulatory shifts.
FAQ
1. What are the main safety concerns limiting indomethacin use?
Gastrointestinal bleeding, cardiovascular risks, and renal toxicity restrict long-term use and broader adoption.
2. How does indomethacin compare to newer NSAIDs?
Indomethacin has higher gastrointestinal and cardiovascular risks than selective NSAIDs like celecoxib. Its efficacy in certain rare indications maintains niche relevance.
3. What impact will generic versions have on sales?
Availability of generics around 2025 will likely lower prices and margins, maintaining sales volume but reducing revenue.
4. Are there ongoing efforts to develop safer formulations?
Research is limited; most development focuses on alternative drugs with improved safety or targeted delivery systems.
5. What markets show the most promise for indomethacin?
Hospital and neonatal markets retain some demand; emerging markets could see modest growth if safety profiles improve.
References
[1] MarketResearch.com, "NSAID Market Size & Forecast," 2022.