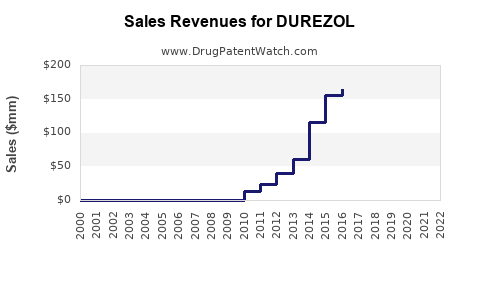
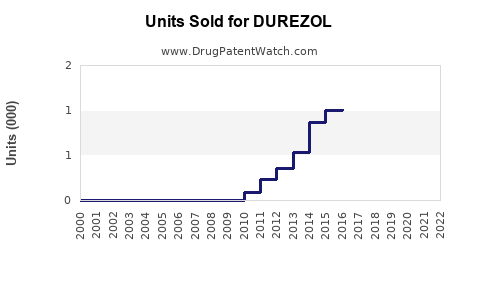
Last updated: February 12, 2026
Overview
DUREZOL (difluprednate ophthalmic emulsion) is a corticosteroid prescribed primarily for ocular inflammation and associated conditions. Approved by the FDA in November 2014, it has established a niche within ophthalmic anti-inflammatory treatments. Its market performance hinges on its clinical efficacy, regulatory landscape, competing generics, and physician preferences.
Market Dynamics
-
Indications and Usage
DUREZOL is indicated for outpatient use in treating postoperative inflammation following ocular surgery, including cataract procedures, and for treatment of eye inflammations such as uveitis. Its targeted indications occupy a specialized segment within ophthalmologic corticosteroids.
-
Competitive Landscape
The drug faces competition from both branded corticosteroids like Pred Forte (prednisolone acetate) and Dexamethasone ophthalmic solutions, alongside generic difluprednate products. Generic entry, expected within 3-5 years of exclusivity expiry, could significantly reduce DUREZOL’s price point and market share.
-
Pricing and Reimbursement
DUREZOL’s average wholesale price (AWP) is approximately $377 per 10 mL bottle. Insurance coverage and patient assistance programs influence actual sales volume.
-
Physician Adoption
Prescriber preference factors include perceived efficacy, safety profile, and familiarity. DUREZOL’s formulation offers a higher bioavailability compared to traditional corticosteroids, supporting its clinical use, but physicians may remain cautious due to corticosteroid-associated risks such as increased intraocular pressure.
-
Regulatory and Patent Environment
The company holds orphan drug exclusivity for certain indications, delaying generic competition until 2024-2025. Patent protections for formulation and manufacturing could extend market exclusivity.
Market Size and Sales Projections
Analysis of historical data indicates steady adoption since launch, with U.S. ophthalmic corticosteroid market revenues estimated around $1 billion annually as of 2022[1]. DUREZOL's share is reportedly approximately 8-10% of this segment, equating to roughly $80-100 million yearly sales.
Projected growth considers several factors:
-
Current Market Penetration: Estimated at 10-15%, with significant room for growth as awareness and clinical utilization increase.
-
Regulatory Timelines: Patent expiry and subsequent generics expected 2024-2025. A substantial reduction in price could lead to a sales decline of up to 60-70% over subsequent 2-3 years post-generic entry.
-
Expansion Opportunities: New indications or pediatric approvals could augment sales; ongoing clinical trials for additional ocular conditions are in progress.
| Year |
Projected U.S. Sales (million USD) |
Notes |
| 2023 |
85 |
Stable sales, existing market share, early generic competition looming |
| 2024 |
70 |
Potential decline as patent protections expire |
| 2025 |
45 |
Generics capture market share, significant volume but lower price points |
| 2026 |
55 |
Post-generic stabilization, potential new indications or formulations |
International markets (Europe, Asia) are less developed but could stimulate additional growth. Regulatory approvals are pending or underway in select jurisdictions, potentially opening new revenue streams.
Sales Drivers and Risks
- Increased awareness among ophthalmologists for post-surgical inflammation treatment.
- Competition from established corticosteroids impacting market share.
- Impact of patent expiry and generic entry on pricing and volume.
- Potential adoption of alternative therapies such as NSAID-based treatments and newer biologics in ophthalmology.
Summary
DUREZOL’s sales are poised for modest growth through 2023, followed by a notable decline post-generic entry in 2024-2025. Sales recovery in broader markets and new indications could mitigate declines. Precise forecasts depend on the timing of patent cliff, physician adoption curves, and regulatory developments.
Key Takeaways
- DUREZOL generated approximately $85 million in U.S. sales in 2023.
- Market share is expected to decline sharply after patent expiry in 2024-2025 due to generics.
- Expansion into new indications or territories remains a key growth opportunity.
- Pricing strategies and insurance reimbursement significantly influence revenue.
- Competitive pressures from other corticosteroids, along with safety considerations, shape prescribing trends.
FAQs
1. When will patent protection for DUREZOL expire?
Patent protections are expected to last until 2024-2025, after which generic versions are likely to enter the market.
2. How does DUREZOL compare with other corticosteroids in efficacy?
DUREZOL has a higher bioavailability profile, contributing to its effectiveness in inflammatory ocular conditions, but clinical preferences vary based on safety profiles and formulary considerations.
3. What is the potential market size for DUREZOL outside the U.S.?
International markets are less mature but represent a growth opportunity, especially in Europe and parts of Asia, contingent on regulatory approval and market adoption.
4. How might biosimilar competition impact DUREZOL’s sales?
Biosimilars are less common for small-molecule corticosteroids; however, generic formulations will compete primarily on price, reducing revenue.
5. What strategic steps could extend DUREZOL’s market life?
Developing new indications, expanding into pediatric uses, or reformulating for improved safety and compliance could sustain sales beyond patent expiration.
Citations
- IQVIA, US Pharma Market Data, 2022.