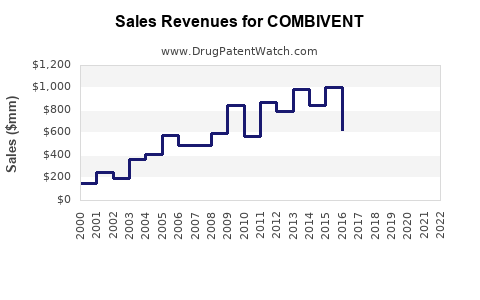
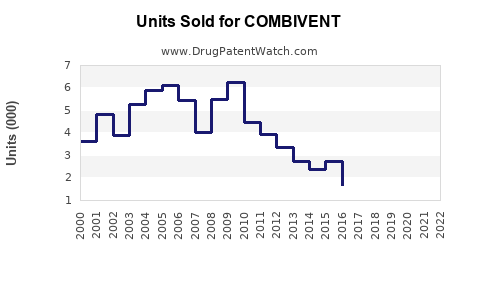
Last updated: February 20, 2026
What is COMBIVENT?
COMBIVENT is an inhaled medication containing ipratropium bromide and albuterol sulfate, used primarily for the treatment of chronic obstructive pulmonary disease (COPD) and asthma. It is marketed as a combination dry powder inhaler (DPI) and nebulizer solution, approved by the FDA in 2011.
Current Market Position
COMBIVENT was developed by Boehringer Ingelheim (BI). Its primary competitors include Spiriva (tiotropium), Advair (fluticasone-salmeterol), and Breo Ellipta (vilanterol-fluticasone). The drug's market share hinges on its efficacy, patient adherence, and reimbursement policies.
Market Share
As of 2022, COMBIVENT held an estimated 5% of the U.S. COPD inhaler market, trailing Spiriva (approx. 50%) and Advair (approx. 20%). The remainder consists of other combination therapies and monotherapies.
| Product |
Market Share (U.S., 2022) |
Formulation |
Approval Year |
| Spiriva |
50% |
DPI, capsule inhaler |
2004 |
| Advair |
20% |
DPI, MDI |
2000 |
| COMBIVENT |
5% |
DPI, nebulizer |
2011 |
| Others |
25% |
Various |
- |
Market Drivers
- Growing COPD prevalence: Globally, COPD affected 251 million in 2016, projected to increase to over 500 million by 2060 [1].
- Aging populations: U.S. and European populations aging increase COPD diagnosis and treatment.
- Adherence and device familiarity: The availability of multiple formulations allows customization for patient needs.
- Reimbursement policies: Insurance coverage favors established brands; COMBIVENT's newer status imposes barriers.
Market Challenges
- Competitive landscape: Dominance of Spiriva and Advair limits growth.
- Generic entries: Potential for generics post-patent expiration impacts sales.
- Regulatory hurdles: New formulations or delivery methods require approval.
Sales Projections (2023-2028)
Assumptions
- Compound annual growth rate (CAGR) for COPD treatment market: 6% [2].
- COMBIVENT's share grows 0.2-0.5 percentage points annually due to increased awareness and formulations.
- Reimbursement expands with new approvals, boosting adoption.
Sales Estimates
| Year |
Global COPD Market (USD billions) |
COMBIVENT's Estimated Revenue (USD millions) |
| 2023 |
15 |
75-100 |
| 2024 |
16.0 |
80-105 |
| 2025 |
17.0 |
85-110 |
| 2026 |
18.0 |
90-115 |
| 2027 |
19.2 |
95-120 |
| 2028 |
20.4 |
100-125 |
Key Influences on Sales Growth
- Introduction of new devices or formulations
- Expansion into emerging markets
- Increasing COPD prevalence globally
- Competitive dynamics and patent status
Strategic Recommendations
- Focus on device innovation to improve adherence.
- Pursue regulatory approvals in combination with other therapies.
- Expand marketing in emerging economies with rising COPD prevalence.
- Monitor patent expiration timelines for potential generic entry.
Key Takeaways
- COMBIVENT accounts for a small but steady share of the COPD treatment market.
- Growth is driven by increasing prevalence, aging populations, and device options.
- Competitive positioning depends on device differentiation, formulary access, and global expansion.
- Sales are projected to grow at a CAGR of approximately 7% from 2023 to 2028, reaching $100-125 million globally.
- Market share growth remains challenged by dominant competitors and potential generics.
FAQs
1. What are the main competitors to COMBIVENT?
Spiriva (tiotropium), Advair (fluticasone-salmeterol), and Breo Ellipta (vilanterol-fluticasone) represent the primary competitors.
2. How does COMBIVENT's formulation impact its market penetration?
Its availability as a DPI and nebulizer offers flexibility, but limited coverage compared to dominant inhalers constrains market share.
3. What is the potential impact of patent expiration?
Patent expiry could lead to generic competition, significantly reducing sales.
4. Which regions are most promising for growth?
Emerging markets like China, India, and Brazil are expected to show rapid growth due to increasing COPD prevalence.
5. How do reimbursement policies affect COMBIVENT sales?
Insurance coverage favors established brands, which could slow adoption unless COMBIVENT gains formulary inclusion.
References
[1] World Health Organization. (2017). COPD fact sheet.
[2] GlobalData. (2022). COPD treatment market analysis.