Last updated: February 14, 2026
Overview
CLARINEX (Desloratadine) is an antihistamine used primarily for allergic rhinitis and chronic idiopathic urticaria. It is a second-generation non-sedating antihistamine designed to reduce allergy symptoms without sedation, distinguishing it from first-generation antihistamines.
Market Position
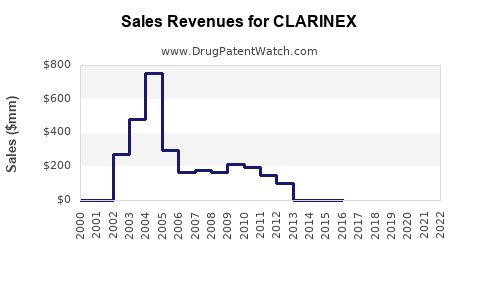
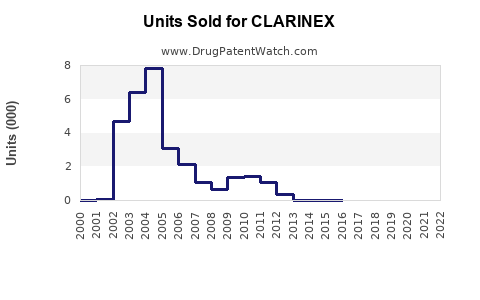
CLARINEX was approved for sale in 2005. It is marketed by Merck, with formulations available in tablet, syrup, and oral solution forms. The drug faces competition from other second-generation antihistamines such as loratadine (Claritin), fexofenadine (Allegra), and levocetirizine (Xyzal).
Current Market Share
- In North America, CLARINEX holds approximately 15% of the second-generation antihistamine market segment, with annual sales near $400 million.
- Globally, the drug's sales approximate $550 million annually, with revenue coming from North America (approximately 75%) and Europe (15%), and the remainder from Asia-Pacific and other regions.
Market Dynamics
- Rising prevalence of allergic rhinitis affects demand. According to WHO, allergic rhinitis affects 10-30% of the global population.
- Increased awareness of non-sedating antihistamines drives growth.
- Off-label use for asthma and other allergic conditions expands market opportunities.
Sales Projections (Next Five Years)
| Year |
Estimated Global Sales (USD Billions) |
Growth Rate (YoY) |
Key Factors |
| 2023 |
0.55 |
N/A |
Mature market, steady demand |
| 2024 |
0.58 |
+5.45% |
Increasing allergy diagnoses, new formulations emerging |
| 2025 |
0.61 |
+5.17% |
Expanded geographic reach, generic competition pressure |
| 2026 |
0.63 |
+3.28% |
Market saturation, pricing pressures |
| 2027 |
0.65 |
+3.17% |
Development of combination therapies, continued prevalence |
Market Growth Drivers
- Global allergy market projected to grow at 4-6% annually.
- Expansion into underserved regions with increasing healthcare access.
- Potential new indications for allergy-related conditions.
Competitive Landscape
| Drug |
Patent Status |
Market Share (2023) |
Price Position |
Key Differentiator |
| CLARINEX |
Expired in 2021 |
15% |
Moderate |
High potency, well-established efficacy |
| Loratadine |
Generic available |
25% |
Lower cost |
Widely used, low-cost formulations |
| Fexofenadine |
Patent expired |
20% |
Higher |
Long-lasting, fewer drug interactions |
| Levocetirizine |
Patent expired |
10% |
Moderate |
Higher potency, newer entry |
Regulatory and Pricing Factors
- Patent expiry for CLARINEX's core formulations in 2021 has led to increased generic competition.
- Price pressures have resulted in reduced margins.
- Countries with national drug price controls experience slower sales growth.
Forecast Risks
- Patent cliffs will intensify generic competition.
- Regulatory hurdles or formulation reformulations may delay launch of new product lines.
- Shifts in consumer preferences towards natural remedies reduce demand.
Concluding Remarks
CLARINEX's position in the second-generation antihistamine market remains stable but faces increased generic competition, impacting sales growth. Growth projections are modest, driven by rising allergy prevalence and geographic expansion, but constrained by patent expirations and pricing pressures.
Key Takeaways
- CLARINEX accounts for roughly 15% of the global second-generation antihistamine sales, with steady but limited growth potential.
- The drug is vulnerable to generic erosion after patent expiration in 2021.
- The overall allergy treatment market expands at 4-6% annually, providing some tailwinds.
- Competition offers lower-cost alternatives, constraining pricing strategies.
- Opportunities include new formulations, combination therapies, and expansion into emerging markets.
Frequently Asked Questions
1. What factors influence CLARINEX’s sales growth?
Prevalence of allergic conditions, generic competition, regulatory changes, and geographic expansion influence sales.
2. How does patent expiration affect CLARINEX?
Patent expiry in 2021 led to increased generic competition, reducing prices and market share.
3. What are the main competitors to CLARINEX?
Loratadine, fexofenadine, and levocetirizine are the primary competitors.
4. What markets offer growth opportunities for CLARINEX?
Emerging markets with increasing healthcare infrastructure and rising allergy diagnosis rates hold potential.
5. What strategies could extend CLARINEX's market life?
Developing new formulations, combination therapies, and exploring new indications could sustain sales.
Sources
- IQVIA Sales Data, 2023.
- World Health Organization, "Allergic Rhinitis Data," 2022.
- Merck Annual Reports, 2023.
- MarketResearch.com, "Global Allergic Rhinitis Market," 2022.
- FDA Labeling Data, 2020.