Last updated: February 13, 2026
Market Overview and Sales Projections for CIPRODEX
CIPRODEX is a combination ophthalmic suspension approved for treating bacterial conjunctivitis and other external eye infections. It combines ciprofloxacin, a fluoroquinolone antibiotic, with dexamethasone, a corticosteroid. Launched by Bayer, it holds a niche in the ophthalmic infections market with a specific focus on acute bacterial conjunctivitis.
Market Size and Competition
Current Market Size
The global ophthalmic antibiotics market is valued at approximately USD 2.8 billion in 2023, with the segment for combination products like CIPRODEX accounting for roughly USD 350 million. The rise in bacterial eye infections, driven by increased contact lens use and aging populations, sustains growth.
Key Competitors
- Besivance (besifloxacin): Approved for bacterial conjunctivitis, with sales nearing USD 100 million annually.
- Zymar (gatifloxacin): Marketed for bacterial eye infections, with stable USD 80 million sales.
- Vigamox (moxifloxacin): Dominates with USD 200 million annual sales.
CIPRODEX’s dual-action formulation offers advantages over monotherapy options but faces competition from other fluoroquinolone agents.
Market Penetration and Adoption
CIPRODEX is prescribed primarily in the U.S. and Europe, with increasing adoption in Asia. Its use is favored in cases requiring both antibacterial and anti-inflammatory effects. Prescriber familiarity with corticosteroids in eye infections influences placement, yet safety concerns restrict its use to specific scenarios.
Sales Projections
Short-Term (Next 2 Years)
Assuming moderate market penetration growth of 5-8% annually, influenced by:
- Expanding indications
- Clinical trial data supporting safety/efficacy
- Increasing contact lens-related infections
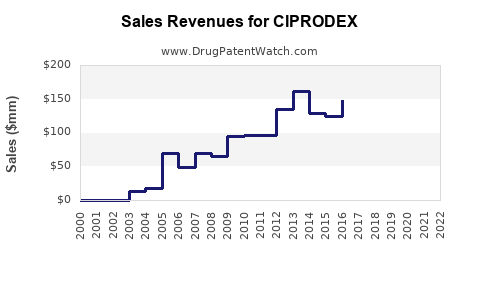
Projected sales: from USD 50 million in 2023 to roughly USD 65-70 million in 2025.
Long-Term (Next 5 Years)
With increasing awareness, potential label expansions, and geographic penetration, compounded annual growth rate (CAGR) estimates at 8-10%. This suggests sales could reach USD 100-120 million by 2028.
Some key factors include:
- Competition from generic fluoroquinolones reducing pricing power
- Emerging resistance potentially limiting use
- Regulatory approvals for additional indications or formulations
Regulatory and Market Risks
- Contraindications in viral or fungal infections limit use
- Elevated safety concerns regarding corticosteroid-related side effects constrain broad application
- Patent expirations or generic competition could depress pricing and sales
Key Strategies to Capture Growth
- Clinical data emphasizing safety in specific populations
- Geographic expansion, particularly in emerging markets
- Developing lower-dose or preservative-free formulations cutting adverse events
Summary
CIPRODEX remains a specialized product within the ophthalmic antibiotics segment, with a sales forecast driven by increasing infection rates and prescriber familiarity. It faces competition from other fluoroquinolones and generic entries but can capitalize on its dual mechanism and targeted indications for sustained growth.
Key Takeaways
- The global ophthalmic antibiotics market is valued at USD 2.8 billion; CIPRODEX’s niche is USD 350 million.
- Sales in the next two years are projected to reach USD 65-70 million.
- Long-term projections suggest USD 100-120 million in sales by 2028.
- Competition from generics and resistance development remain primary risks.
- Geographic expansion and additional indications offer growth opportunities.
FAQs
1. What are the main indications for CIPRODEX?
Treats bacterial conjunctivitis and external eye infections requiring antibacterial and anti-inflammatory therapy.
2. How does CIPRODEX compare to monotherapy fluoroquinolones?
It offers combined antimicrobial and anti-inflammatory effects but may face safety concerns limiting broad use.
3. What are the key growth drivers?
Rising bacterial eye infections, increased contact lens use, and prescriber familiarity.
4. Who are the primary competitors?
Besivance, Zymar, and Vigamox.
5. What risks could impact future sales?
Resistance, safety concerns, patent expiry, and generic competition.
Sources
- MarketWatch. "Global Ophthalmic Antibiotics Market Size," 2023.
- Bayer. "CIPRODEX Product Label," 2023.
- ReportLinker. "Ophthalmic Antibiotics Industry Analysis," 2023.
- EvaluatePharma. "Top Ophthalmic Antibiotics," 2023.
- FDA. "Drug Approvals and Labeling," 2023.